FDA Grants IND Approval to Promising Anti-Cancer Agent Veyonda®
25 Febbraio 2020 - 2:30PM
Business Wire
Current IND for sarcoma paves the way for
future INDs for late-stage prostate cancer
Noxopharm (ASX: NOX) announced today that the U.S. Food and Drug
Administration (FDA) has approved the Investigational New Drug
(IND) application for Veyonda® for combination treatment with
doxorubicin in patients with soft tissue sarcomas.
“Based on preclinical and clinical data, the FDA’s approval of
this IND is validation of the clinical potential of Veyonda®,” said
Noxopharm CMO Gisela Mautner, M.D., Ph.D. “Further, it means that
Veyonda® has met stringent FDA standards for safety and
tolerability.”
“While the Company’s resources and energies are primarily
focused on bringing a treatment for late-stage prostate cancer to
market, this IND grant for a less common cancer type advances that
commercial objective,” said Noxopharm CEO Graham Kelly, Ph.D. “The
recent IND approval process has familiarized the FDA with Veyonda®
ahead of IND applications for more advanced DARRT and LuPIN study
submissions. It also opens the door to Veyonda®’s use in the U.S.,
where patient demand has been growing.”
DARRT and LuPIN are ongoing investigations in which combination
therapies with Veyonda® are exhibiting promising interim results
for the treatment of late-stage, progressive prostate cancer
resistant to other treatments.
“This IND grant is an important step toward achieving a
strategic partnering arrangement,” Kelly said. “With the emerging
clinical data and growing evidence that Veyonda® is a uniquely
acting immuno-oncology drug candidate designed to restore immune
function to cold tumors, we are confident that the industry will
recognize the considerable opportunities in the adjunctive benefits
of Veyonda®.”
Veyonda® is a suppository dosage form of idronoxil, a
first-in-class inhibitor of sphingosine-1-phosphate (S1P). By
inhibiting the overexpression of S1P-driven pro-survival pathways
in solid tumors, idronoxil is designed to sensitize cancer cells to
the anti-cancer effects of drugs such as doxorubicin, as well as to
radiation.
About Noxopharm Noxopharm is a clinical-stage Australian
oncology drug development company with offices in Sydney and New
York. The Company has a primary focus on the development of
Veyonda® and is the major shareholder in the non-oncology drug
development company, Nyrada Inc. (ASX: NYR). Learn more at
noxopharm.com.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20200225005215/en/
Jane Byram SCORR Marketing (512) 626-2758
jane@scorrmarketing.com
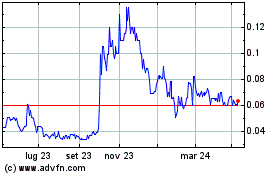
Grafico Azioni Noxopharm (ASX:NOX)
Storico
Da Mar 2024 a Apr 2024
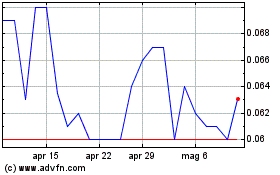
Grafico Azioni Noxopharm (ASX:NOX)
Storico
Da Apr 2023 a Apr 2024