GlaxoSmithKline, SK Bioscience Begin Phase 3 Trial of Covid-19 Vaccine
31 Agosto 2021 - 8:50AM
Dow Jones News
By Adam Clark
GlaxoSmithKline PLC said Tuesday that it and South Korean
company SK Bioscience Co Ltd. have started a Phase 3 trial of their
Covid-19 vaccine combination.
The British pharmaceutical company said the advance follows
positive immunogenicity and safety data from earlier trials and
that the partners are aiming for a global supply of the vaccine via
the Covax facility in the first half of 2022, subject to data and
regulatory review.
The trial combines SK Bioscience's vaccine candidate with GSK's
adjuvant, an ingredient used to prompt a stronger immune
response.
The randomized trial will enroll around 4,000 participants and
compare the vaccine against the AstraZeneca PLC vaccine, with data
due in the first half of 2022.
Write to Adam Clark at adam.clark@wsj.com
(END) Dow Jones Newswires
August 31, 2021 02:35 ET (06:35 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
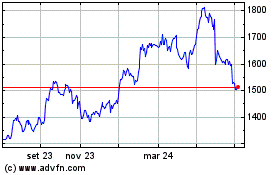
Grafico Azioni Gsk (LSE:GSK)
Storico
Da Mar 2024 a Apr 2024
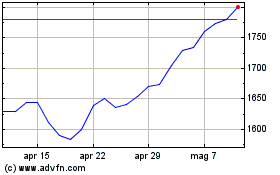
Grafico Azioni Gsk (LSE:GSK)
Storico
Da Apr 2023 a Apr 2024