Guerbet: Gadopiclenol - positive results for Phase III clinical trials
24 Marzo 2021 - 8:00AM

Guerbet: Gadopiclenol - positive results for Phase III clinical
trials
Gadopiclenol: positive results for Phase
III clinical trials
- Gadopiclenol is a high-relaxivity macrocyclic contrast
agent used for magnetic resonance imaging (MRI).
- Its diagnostic efficacy and safety have been evaluated in two
Phase III clinical studies with a dose of gadolinium reduced
by half compared to products already available on the
market.
- Guerbet is planning to make regulatory submissions from early
2022 in a wide range of indications in adults and children
from 2 years of age.
Villepinte (France), March 24, 2021 (8 a.m.
CET) – Guerbet (FR0000032526 GBT), a world leader in
medical imaging, today announced positive results from two Phase
III clinical studies comparing the diagnostic efficacy and safety
of Gadopiclenol to Gadobutrol in a wide range of indications,
covering the central nervous system and various other anatomical
areas (head and neck, thorax, abdomen, pelvis, musculoskeletal
system).
A total of 560 patients were recruited for these
two studies in over 60 hospital medical imaging departments in 13
countries.
The endpoints were met in terms of the
diagnostic benefit of injecting Gadopiclenol (0.05 mmol/kg)
during MRI examinations, based on: 1/ the superiority of the
examination with Gadopiclenol compared to the examination with no
contrast agent; and 2/ the non-inferiority of Gadopiclenol
(0.05 mmol/kg) compared to Gadobutrol (0.1 mmol/kg) for
the visualization and detection of lesions of the central nervous
system and in the other anatomical areas studied.
In addition to these Phase III trials, a study
was conducted on 80 children between 2 and 17 years of age in 19
centers in five European countries. This study showed that the
pharmacokinetic profile of Gadopiclenol at 0.05 mmol/kg in
children was similar to that of adults. It will therefore be
possible to extrapolate the efficacy of Gadopiclenol to the
pediatric population based on data collected in adult patients.
No major safety signals were reported during the
development of Gadopiclenol, and the adverse reactions reported
during the two Phase III studies were similar for both of the
products administered.
“We are very happy with the results of these
studies, which demonstrate the diagnostic value and good tolerance
of Gadopiclenol during an MRI examination in adults and children.
By using a lower dose of gadolinium in clinical practice, we hope
this macrocyclic contrast agent will become a ‘best-in-class’
diagnostic solution for doctors and their patients.” said David
Hale, CEO of Guerbet.
All this data will serve as a basis for
regulatory submissions, which will begin in the United States and
in the European Union (EU) early 2022.
About the
Phase III clinical trials
Details on these two clinical trials are
available in the ClinicalTrials.gov database:
- Efficacy and Safety of Gadopiclenol for Central Nervous System
(CNS) Magnetic Resonance Imaging (MRI) - Full Text View -
ClinicalTrials.gov
- Efficacy and Safety of Gadopiclenol for Body Magnetic Resonance
Imaging (MRI) - Full Text View - ClinicalTrials.gov
About
Gadopiclenol
Gadopiclenol is a new macrocyclic
gadolinium-based contrast agent with high relaxivity (contrast
power), designed and developed by Guerbet’s R&D team. The
efficacy and safety of Gadopiclenol have been evaluated as part of
the company’s clinical development plan with a view to obtaining
worldwide marketing authorization. No regulatory authority has
evaluated the study data for this product to date.
About
Guerbet
Guerbet is a leader in medical imaging
worldwide, offering a wide range of pharmaceutical products,
medical devices, digital and AI solutions for diagnostic and
interventional imaging, to improve the diagnosis and treatment of
patients. A pioneer since 95 years in the field of contrast media
with over 2,600 people globally, Guerbet is continuously innovating
with 10% of revenue dedicated to Research & Development and
four centers in France, Israel and the United States. Guerbet (GBT)
is listed on Euronext Paris (segment B – mid caps) and generated
€712 million in revenue in 2020. For more information about
Guerbet, please visit www.guerbet.com.
Media
relations
Guerbet GlobalAlize RPCaroline
Carmagnol / +33 (0) 6 64 18 99 59 / guerbet@alizerp.com
- Guerbet Press Release - Gadopiclenol - March 24 2021
Grafico Azioni Guerbet (EU:GBT)
Storico
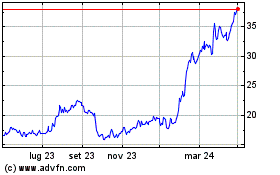
Da Mar 2024 a Apr 2024
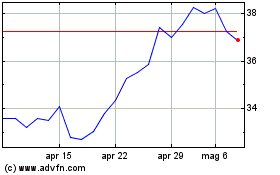
Grafico Azioni Guerbet (EU:GBT)
Storico
Da Apr 2023 a Apr 2024