Nyxoah announces the publication of first positive clinical data in an OSA patient suffering from Complete Concentric Collaps...
03 Marzo 2021 - 6:58AM

Nyxoah announces the publication of first positive clinical data in
an OSA patient suffering from Complete Concentric Collapse (CCC)
PRESS RELEASE
Nyxoah announces the publication of first
positive clinical data in an OSA patient suffering from Complete
Concentric Collapse (CCC)
Mont-Saint-Guibert, Belgium – 3rd March
2021 – Nyxoah SA (Euronext: NYXH) (“Nyxoah” or the
“Company”), a health-technology company focused on the development
and commercialization of innovative solutions and services to treat
Obstructive Sleep Apnea (OSA), today announces the publication of
an article presenting the first case of successful treatment of an
OSA patient suffering from Complete Concentric Collapse (CCC) with
Nyxoah’s unique Genio® bilateral neurostimulation solution1.
Patients suffering from Complete Concentric
Collapse (CCC) currently amount for at least 30% of the existing
OSA patient population. For now, these patients are
contra-indicated for existing Hypoglossal Nerve Stimulation OSA
therapies. Nyxoah is currently conducting the BETTER SLEEP study,
that aims to assess the effectiveness of the Genio® system
bilateral hypoglossal nerve stimulation for Complete Concentric
Collapse (CCC) patients and to build clinical evidence for a
potential therapeutic indication expansion.
Dr. Richard Lewis, leading author of
this publication and Principal Investigator of the BETTER SLEEP
study commented: “This case demonstrates
successful control of obstructive sleep apnea in a patient with
Complete Concentric Collapse (CCC) of the redropalatal
airway. Whilst the final results of the Better Sleep trial are
yet to be completed, the results so far in the CCC patients
indicate that the Genio® system could potentially be a treatment
for OSA patients with CCC who are intolerant of CPAP.”
Olivier Taelman, Chief Executive Officer
of Nyxoah, added: “I would like to thank Dr. Lewis and the
Perth team for getting this manuscript published, since this is the
first and promising step towards offering hypoglossal nerve
stimulation to a cohort of OSA patients who are currently excluded
from such therapy. We will be announcing the full results of our
BETTER SLEEP study in May 2021.”
- ENDS -
For further information, please contact:
Nyxoah
Milena Venkova, Corporate Communications
Manager
milena.venkova@nyxoah.com
+32 490 11 93 57
About Nyxoah
Nyxoah is a healthtech company focused on the
development and commercialization of innovative solutions and
services to treat Obstructive Sleep Apnea (OSA). Nyxoah’s lead
solution is the Genio® system, a CE-validated, patient-centered,
next generation hypoglossal neurostimulation therapy for OSA, the
world’s most common sleep disordered breathing condition that is
associated with increased mortality risk2 and comorbidities
including cardiovascular diseases, depression and stroke.
Following the successful completion of the BLAST
OSA study in patients with moderate to severe OSA, the Genio®
system received its European CE Mark in 2019. The Company is
currently conducting the BETTER SLEEP study in Australia and New
Zealand for therapy indication expansion, the DREAM IDE pivotal
study for FDA approval and a post-marketing EliSA study in Europe
to confirm the long-term safety and efficacy of the Genio®
system.
For more information, please visit
www.nyxoah.com.
Caution – CE marked since 2019.
Investigational device in the United States. Limited by U.S.
federal law to investigational use in the United States.
1 Lewis R, Le J, Czank C, Raux G. Control of OSA in a patient
with CCC of soft palate using bilateral hypoglossal nerve
stimulation. Clin Case Rep. 2021;00:1–3 LINK
2 Young T. et al: Sleep Disordered Breathing and Mortality:
Eighteen-Year Follow-up of the Wisconsin Sleep Cohort, Sleep. 2008
Aug 1; 31(8): 1071–1078
- First positive clinical data in an OSA CCC patient
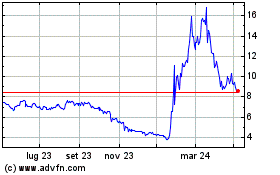
Grafico Azioni Nyxoah (EU:NYXH)
Storico
Da Mar 2024 a Apr 2024
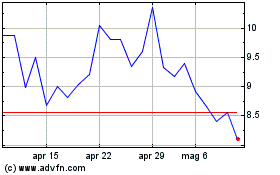
Grafico Azioni Nyxoah (EU:NYXH)
Storico
Da Apr 2023 a Apr 2024