Nyxoah receives FDA approval for full-body 1.5T and 3T MRI compatibility for the Genio® system to treat Obstructive Sleep Ap...
09 Febbraio 2021 - 7:00AM

Nyxoah receives FDA approval for full-body 1.5T and 3T MRI
compatibility for the Genio® system to treat Obstructive Sleep
Apnea (OSA)
PRESS RELEASE
Nyxoah receives FDA approval for
full-body 1.5T and 3T MRI compatibility for the Genio®
system to treat Obstructive Sleep Apnea
(OSA)
Mont-Saint-Guibert, Belgium – 9th
February 2021 – Nyxoah SA (Euronext: NYXH) (“Nyxoah” or
the “Company”), a health-technology company focused on the
development and commercialization of innovative solutions and
services to treat Obstructive Sleep Apnea (OSA), today announces
that the Company has received approval by the Food and Drug
Administration (FDA) for the Magnetic Resonance Imaging (MRI)
conditional labeling for the Genio® neurostimulation-based OSA
therapy, currently being evaluated in the DREAM pivotal IDE
study.
This revised labeling ensures that patients who
receive the Genio® system and those already implanted can now
undergo full-body 1.5T and 3T MRI diagnostic scans within approved
parameters, and access the benefits of Genio® unique bilateral
stimulation therapy.
The DREAM (Dual-sided Hypoglossal neRvE
stimulAtion for the treatMent of Obstructive Sleep Apnea) study is
an Investigational Device Exemption (IDE) trial designed to support
the marketing authorization of the Genio® system in the United
States. This is a multicenter study being conducted worldwide
including sites in the United States, Germany, Belgium and
Australia.
Olivier Taelman, Chief Executive Officer
of Nyxoah, commented: “The approval by the Food and Drug
Administration (FDA), received only a week after similar CE mark
approval in Europe, confirms again the unique and unparalleled
design of our technology. With the prevalence of MRI scans in the
United States being one of the highest in the world, we are
delighted that Nyxoah will be able to fulfil the currently unmet
need for full-body 1.5T and 3T MR conditional labeling. Such an
extensive labeling is unique to Nyxoah in the field of
neurostimulation-based OSA therapies. Currently other therapies
cannot fully address this need due to limitations to 1.5T MRI scans
and body areas exclusion. As a company, Nyxoah always puts the
patient first and seeks to ensure minimal disruption of their daily
life and optimal Quality of Life (QOL).”
Prof. B. Tucker Woodson, MD,
added: “As the Principal Investigator for the
DREAM pivotal IDE study, I’m really pleased with the FDA approval
for full-body 1.5T and 3T MRI compatibility for the Genio® system.
MRI scans are often the preferred diagnostic
imaging modality for comorbidities affecting OSA
patients. This extensive MRI labeling will be a major benefit for
all OSA patients who currently receive the Genio® therapy in the
United States as part of the DREAM IDE clinical trial, ensuring
that they can undergo MRI scans in full safety.”
- ENDS -
For further information, please contact:
NyxoahMilena Venkova, Corporate
Communications Managermilena.venkova@nyxoah.com+32 490 11 93
57
About Nyxoah
Nyxoah is a healthtech company focused on the
development and commercialization of innovative solutions and
services to treat Obstructive Sleep Apnea (OSA). Nyxoah’s lead
solution is the Genio® system, a CE-validated, patient-centered,
next generation hypoglossal neurostimulation therapy for OSA, the
world’s most common sleep disordered breathing condition that is
associated with increased mortality risk1 and comorbidities
including cardiovascular diseases, depression and stroke. Following
the successful completion of the BLAST OSA study in patients with
moderate to severe OSA, the Genio® system received its European CE
Mark in 2019. The Company is currently conducting the BETTER SLEEP
study in Australia and New Zealand for therapy indication
expansion, the DREAM IDE pivotal study for FDA approval and a
post-marketing EliSA study in Europe to confirm the long-term
safety and efficacy of the Genio® system. For more information,
please visit www.nyxoah.com.
Caution – CE marked since 2019.
Investigational device in the United States. Limited by U.S.
federal law to investigational use in the United States.
1 Young T. et al: Sleep Disordered Breathing and Mortality:
Eighteen-Year Follow-up of the Wisconsin Sleep Cohort, Sleep. 2008
Aug 1; 31(8): 1071–1078.
- FDA approval Genio MRI Compatible labeling
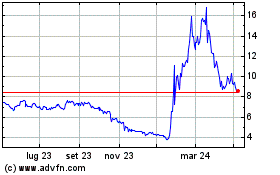
Grafico Azioni Nyxoah (EU:NYXH)
Storico
Da Mar 2024 a Apr 2024
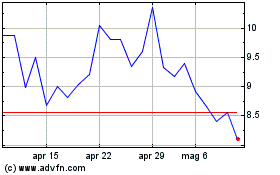
Grafico Azioni Nyxoah (EU:NYXH)
Storico
Da Apr 2023 a Apr 2024