OXURION Confirms Institutional Review Board Approval and Submission of the Investigational New Drug Application to the FDA to...
10 Giugno 2021 - 7:00AM

OXURION Confirms Institutional Review Board Approval and Submission
of the Investigational New Drug Application to the FDA to Start
Phase 2 Study Evaluating THR-687 for Diabetic Macular Edema (DME)
THR-687
is a potent pan-RGD
integrin
antagonistholding
potential as next generation
first line therapy for
DME
Leuven,
BE, Boston, MA,
US – June
10,
2021 –
07.00
AM CET – Oxurion NV (Euronext
Brussels: OXUR), a biopharmaceutical company developing next
generation standard-of-care ophthalmic therapies, with a clinical
stage portfolio in vascular retinal disorders, announces that it
has received Institutional Review Board (IRB) approval to initiate
a Phase 2 clinical study of THR-687 in patients with Diabetic
Macular Edema (study name “INTEGRAL”). Together with its earlier
submission to the U.S. Food and Drug Administration (FDA) of the
final protocol to the Investigational New Drug (IND) Application,
this is an important step forward for the Phase 2 INTEGRAL
study.
Tom Graney, CFA, Chief Executive
Officer of Oxurion,
comments: “Together with our earlier submission to the FDA of the
final protocol to the IND, IRB approval for the THR-687 Phase 2
trial in DME is an important milestone for OXURION. This follows
the completion of enrolment of Part A in our Phase 2 program
evaluating THR-149 in DME as we announced earlier this week. The
team is now ready to also start this second Phase 2 program, and we
are looking forward to working alongside a large team of
enthusiastic investigators across the US and Europe, together
creating new paths towards improved therapies for treatment of DME,
a very important area of unmet medical need in diabetic eye
disease.”
Today’s announcement follows the positive data
reported from a Phase 1, open-label, multi-center (US), single dose
escalation study (n=12) evaluating the safety of a single
intravitreal injection of 3 increasing doses (0.4 mg, 1.0 mg,
2.5 mg) of THR-687 for the treatment of DME.
A single injection of THR-687 was reported safe
and well-tolerated, showing a very encouraging efficacy signal.
Across all doses, a rapid onset of action in mean BCVA was observed
from Day 1 with an increase of 3.1 letters, which further improved
to 9.2 letters at Month 1. This activity was maintained with a mean
BCVA improvement of 8.3 letters at Month 3 following a single
injection of THR-687.
A clear dose response was seen with the greatest
positive effect on BCVA and Central Subfield Thickness (CST) with
the highest dose of THR-687. For this highest dose, a mean
BCVA Improvement of 11 letters was noted at Day 14, with a peak
improvement of 12.5 letters at Month 3. Similarly, a peak mean CST
decrease of 106 µm was observed at Day 14 with the highest dose of
THR-687.
Beyond DME, THR-687 also has development
possibilities in additional vascular retinal disorders including
for wet Age-related Macular Degeneration (wet AMD) and retinal vein
occlusion (RVO), thereby potentially allowing the Company to tap
into a broader therapeutic market with a current combined estimated
annual value of $12+ billion.
END
For further information please
contact:
|
Oxurion NVWouter Piepers, Global Head of Investor Relations&
Corporate CommunicationsTel: +32 16 75 13 10 / +32 478 33 56
32wouter.piepers@oxurion.com |
EU Citigate Dewe Rogerson David Dible/ Sylvie Berrebi/Frazer
HallTel: +44 20 7638
9571oxurion@citigatedewerogerson.com USWestwicke, an ICR
CompanyChristopher BrinzeyTel: +1 617 835
9304chris.brinzey@westwicke.com |
About Oxurion
Oxurion (Euronext Brussels: OXUR) is a
biopharmaceutical company developing next generation standard of
care ophthalmic therapies, which are designed to better preserve
vision in patients with retinal vascular disorders including
diabetic macular edema (DME), the leading cause of vision loss in
diabetic patients worldwide as well as other conditions, including
wet age-related macular degeneration (AMD) and retinal vein
occlusion (RVO).
Oxurion is aiming to build the leading global
franchise in the treatment of retinal vascular disorders based on
the successful development of its two novel therapeutics:
- THR-149 is a plasma kallikrein
inhibitor being developed as a potential new standard of care for
the 40% of DME patients who respond suboptimally to anti-VEGF
therapy. THR-149 has shown positive topline Phase 1 results for the
treatment of DME. The company is currently conducting a Phase 2
clinical trial evaluating multiple injections of THR-149 in DME
patients who previously responded suboptimally to anti-VEGF
therapy.
- THR-687 is a pan-RGD integrin
antagonist that is initially being developed as a potential first
line therapy for DME patients. Positive topline results in a Phase
1 clinical study assessing THR-687 as a treatment for DME were
announced in 2020. THR-687 is expected to enter a Phase 2 clinical
trial in mid-2021. THR-687 also holds the potential to deliver
improved treatment outcomes for patients with wet AMD and RVO.
Oxurion is headquartered in Leuven, Belgium, and
is listed on the Euronext Brussels exchange under the symbol OXUR.
More information is available at www.oxurion.com.
Important information about
forward-looking statements
Certain statements in this press release may be
considered “forward-looking”. Such forward-looking statements are
based on current expectations, and, accordingly, entail and are
influenced by various risks and uncertainties. The Company
therefore cannot provide any assurance that such forward-looking
statements will materialize and does not assume an obligation to
update or revise any forward-looking statement, whether as a result
of new information, future events, or any other reason. Additional
information concerning risks and uncertainties affecting the
business and other factors that could cause actual results to
differ materially from any forward-looking statement is contained
in the Company’s Annual Report. This press release does not
constitute an offer or invitation for the sale or purchase of
securities or assets of Oxurion in any jurisdiction. No
securities of Oxurion may be offered or sold within the United
States without registration under the U.S. Securities Act of 1933,
as amended, or in compliance with an exemption therefrom, and in
accordance with any applicable U.S. state securities laws.
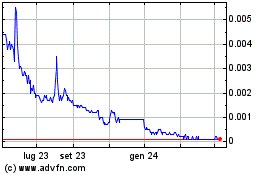
Grafico Azioni Oxurion NV (EU:OXUR)
Storico
Da Mar 2024 a Apr 2024
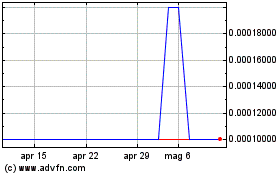
Grafico Azioni Oxurion NV (EU:OXUR)
Storico
Da Apr 2023 a Apr 2024