Première implantation sur un patient américain du système Genio® pour le traitement du Syndrome d’Apnées Obstructives ...
05 Gennaio 2021 - 7:39AM

Première implantation sur un patient américain du système Genio®
pour le traitement du Syndrome d’Apnées Obstructives du Sommeil
(SAOS) dans le cadre de l’étude pivot IDE DREAM
PRESS RELEASE
First US patient implanted in the DREAM pivotal IDE study,
with the Genio® system for the treatment of Obstructive Sleep Apnea
(OSA)
DREAM is a pivotal, Investigational Device Exemption (IDE) study,
designed to support marketing authorization in the United
States
Mont-Saint-Guibert, Belgium – 5th January 2021 –
Nyxoah S.A. (EBR: NYXH) (“Nyxoah” or the “Company”), a
health-technology company focused on the development and
commercialization of innovative solutions and services to treat
Obstructive Sleep Apnea (OSA), today announces the successful
implantation of the first US patient in the DREAM pivotal IDE
study. The implantation took place at the Nose and Sinus Institute
Boca Raton, Florida and was performed by Dr. Melyssa Hancock,
Otolaryngology-Head & Neck surgeon.
The DREAM (Dual-sided Hypoglossal neRvE stimulAtion for the
treatMent of Obstructive Sleep Apnea) study is a pivotal,
Investigational Device Exemption (IDE) trial designed to support
the marketing authorization of the Genio® system in the United
States. This multicenter, prospective, open-label,
observational study will enroll 134 patients, who will undergo the
implantation procedure in up to 26 centers worldwide including
sites in the United States, Germany, Belgium and Australia.
Dr. Melyssa Hancock, implanting surgeon from the Nose and
Sinus Institute Boca Raton commented: “ We are very
excited to be chosen as the first center in the United States to
implant the Genio system in a patient for treatment of obstructive
sleep apnea. The teaming of the Nose and Sinus Institute of Boca
Raton with the innovators at Nyxoah represents the collaboration of
some of the most experienced surgeons in the United States today
treating nose and airway issues with a team of brilliant
international engineers. During this time of COVID-19 and virtual
adaptation to everything we do, the fact that we were able to
communicate in real-time during the procedure with other surgeons
globally who have extensive experience with this device made it a
truly extraordinary and successful endeavor.”
Olivier Taelman, Chief Executive Officer of Nyxoah,
added: “I’m really proud of Nyxoah’s team reaching another
key milestone despite all challenges due to the Covid-19 pandemic
and would like to congratulate Dr. Hancock and her team for
implanting the first US patient with the Genio® system.
Enabling US physicians to build their experience with the Genio®
system, combined with the existing expertise of other international
surgeons participating in the DREAM study, is supporting Nyxoah’s
timeline for the pivotal IDE study enrollment closing by the end of
Q2 2021.
- ENDS -
For further information, please contact:
Nyxoah
Milena Venkova, Corporate Communications Manager
milena.venkova@nyxoah.com
+32 490 11 93 57
About Nyxoah
Nyxoah is a healthtech company focused on the development and
commercialization of innovative solutions and services to treat
Obstructive Sleep Apnea (OSA). Nyxoah’s lead solution is the Genio®
system, a CE-validated, patient-centered, next generation
hypoglossal neurostimulation therapy for OSA, the world’s most
common sleep disordered breathing condition that is associated with
increased mortality risk1 and comorbidities including
cardiovascular diseases, depression and stroke.
Following the successful completion of the BLAST OSA study in
patients with moderate to severe OSA, the Genio® system received
its European CE Mark in 2019. The Company is currently conducting
the BETTER SLEEP study in Australia and New Zealand for therapy
indication expansion, the DREAM IDE pivotal study for FDA approval
and a post-marketing EliSA study in Europe to confirm the long-term
safety and efficacy of the Genio® system.
For more information, please visit www.nyxoah.com.
Caution – CE marked since 2019. Investigational
device in the United States. Limited by U.S. federal law to
investigational use in the United States.
1 Young T. et al: Sleep Disordered Breathing and Mortality:
Eighteen-Year Follow-up of the Wisconsin Sleep Cohort, Sleep. 2008
Aug 1; 31(8): 1071–1078.
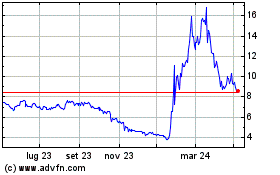
Grafico Azioni Nyxoah (EU:NYXH)
Storico
Da Mar 2024 a Apr 2024
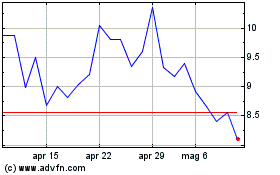
Grafico Azioni Nyxoah (EU:NYXH)
Storico
Da Apr 2023 a Apr 2024