Avacta’s Proprietary TMAC Cancer Therapy Platform Demonstrates Successful Proof-of-Concept in Pre-Clinical Study
28 Gennaio 2020 - 2:27PM
Business Wire
Positive in vivo efficacy and pharmacokinetic
data shows AVA04-VbP drug conjugate out-performed Bavencio
immunotherapy
Avacta Group plc (AIM: AVCT), the developer of Affimer®
biotherapeutics and reagents, is pleased to announce that it has
demonstrated initial proof-of-concept for its proprietary new class
of drug conjugate, TMAC™, in a pre-clinical animal model of cancer.
The study showed that AVA04-VbP outperformed Bavencio (Avelumab), a
marketed anti-PD-L1 immunotherapy.
Avacta’s tumour microenvironment activated drug conjugates
(TMAC) are a ground-breaking new form of cancer immunotherapy,
combining its Affimer biotherapeutics with chemotherapies in a
single drug, using a linker (incorporating its proprietary
pre|CISION™ substrate) that is designed to only release the
chemotherapy in the tumour microenvironment through the action of
an extracellular enzyme called FAPα. This mechanism overcomes the
need to target an internalising cancer marker, as with conventional
antibody drug conjugates (ADCs), and allows extremely potent
chemotherapies to be combined with Affimer immune-checkpoint
therapies. Combining pre|CISION technology with the Affimer
platform is enabling Avacta to build a pipeline of novel and safe
cancer therapies applicable to a broader range of cancer patients,
including those who do not respond to existing immunotherapies.
AVA04-VbP combines an Affimer PD-L1 checkpoint inhibitor with an
I-DASH chemotherapy warhead, addressing the acute systemic toxicity
associated with I-DASH inhibitors by targeting the release of the
drug warhead in the tumour microenvironment. The drug warhead
induces a highly pro-inflammatory cell death, which in turn
stimulates an immune response in the tumour, which is supported by
the Affimer PD-L1/PD-1 signalling pathway blockade.
In a mouse syngeneic tumour model study, Avacta has shown that
AVA04-VbP outperforms Bavencio; animals treated with AVA04-VbP
showed a significant reduction in the rate of tumour growth with
respect to those treated with Bavencio. A considerably higher level
of the released I-DASH warhead was measured in the tumours compared
with very low levels in the blood, indicating that healthy tissues
are less exposed to the highly toxic warhead. This tumour targeting
is central to the TMAC mechanism of action, and permits the use of
the highly potent cancer-killing warheads.
Dr Alastair Smith, Chief Executive Officer of Avacta Group,
commented: “These in vivo efficacy and pharmacokinetic data
demonstrate initial proof-of-concept for TMAC drug conjugates and
represent an important milestone for the Company.” He added:
“We have received significant commercial interest in the TMAC
concept, so I am delighted that we have been able to demonstrate
superior efficacy and targeting of the warhead to the tumour with
the first TMAC to be have tested in animals. This is hugely
encouraging for the TMAC programme and I look forward to keeping
the market updated on future developments.”
About Avacta Group plc - https://www.avacta.com
View source
version on businesswire.com: https://www.businesswire.com/news/home/20200128005454/en/
Zyme Communications (Trade and Regional Media) Katie
Odgaard Tel: +44 (0)7787 502 947
katie.odgaard@zymecommunications.com
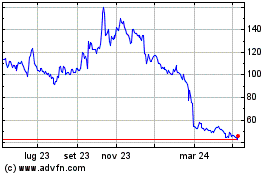
Grafico Azioni Avacta (LSE:AVCT)
Storico
Da Mar 2024 a Apr 2024
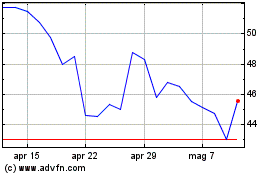
Grafico Azioni Avacta (LSE:AVCT)
Storico
Da Apr 2023 a Apr 2024