Form 8-K - Current report
11 Marzo 2024 - 11:04AM
Edgar (US Regulatory)
false
0001855644
0001855644
2024-03-11
2024-03-11
0001855644
us-gaap:CommonClassAMember
2024-03-11
2024-03-11
0001855644
us-gaap:WarrantMember
2024-03-11
2024-03-11
iso4217:USD
xbrli:shares
iso4217:USD
xbrli:shares
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
Current Report
Pursuant to Section 13 or 15(d) of the
Securities Exchange Act of 1934
March 11, 2024
Date of Report (Date of earliest event reported)
Zura
Bio Limited
(Exact Name of Registrant
as Specified in its Charter)
| Cayman Islands |
|
001-40598 |
|
98-1725736 |
(State or other jurisdiction
of incorporation) |
|
(Commission
File Number) |
|
(I.R.S. Employer
Identification No.) |
| 1489 W. Warm Springs Rd. #110 |
|
|
| Henderson, Nevada |
|
89014 |
| (Address of Principal Executive Offices) |
|
(Zip Code) |
Registrant's telephone number,
including area code: (702) 757-6133
N/A
(Former name or former address,
if changed since last report)
Check the appropriate box below if the Form 8-K
filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| ¨ | Written
communications pursuant to Rule 425 under the Securities Act |
| ¨ | Soliciting
material pursuant to Rule 14a-12 under the Exchange Act |
| ¨ | Pre-commencement
communications pursuant to Rule 14d-2(b) under the Exchange Act |
| ¨ | Pre-commencement
communications pursuant to Rule 13e-4(c) under the Exchange Act |
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class |
|
Trading
Symbol(s) |
|
Name
of each
exchange on
which registered |
| Class A Ordinary Shares, par value $0.0001 per share |
|
ZURA |
|
The Nasdaq Stock Market |
| Warrants, each whole warrant exercisable for one Class A Ordinary Share at an exercise price of $11.50 per share |
|
ZURAW |
|
The Nasdaq Stock Market |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (17 CFR §230.405) or Rule 12b-2 of the Securities Exchange Act of 1934 (17 CFR §240.12b-2).
Emerging growth company x
If an emerging growth company, indicate by check mark if the registrant
has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant
to Section 13(a) of the Exchange Act.
| Item 7.01 |
Regulation FD Disclosure. |
On March
11, 2024, representatives of Zura Bio Limited, a Cayman Islands exempted company (the “Company”), began making presentations
to banks and analysts using slides containing the information attached to this Current Report on Form 8-K as Exhibit 99.1 (the “Investor
Presentation”), which is incorporated herein by reference. The Company has filed the Investor Presentation on its website and expects
to use the Investor Presentation, in whole or in part, and possibly with modifications, in connection with presentations to investors,
analysts and others during the fiscal year ending December 31, 2024.
By filing
this Current Report on Form 8-K and furnishing the information contained herein, the Company makes no admission as to the materiality
of any information in this report that is required to be disclosed solely by reason of Regulation FD.
The information
contained in the Investor Presentation is summary information that is intended to be considered in the context of the Company’s
Securities and Exchange Commission (“SEC”) filings and other public announcements that the Company may make, by press release
or otherwise, from time to time. The Company undertakes no duty or obligation to publicly update or revise the information contained in
this report, although it may do so from time to time as its management believes is warranted. Any such updating may be made through the
filing of other reports or documents with the SEC, through press releases or through other public disclosure.
The information
presented in Item 7.01 of this Current Report on Form 8-K and Exhibit 99.1 shall not be deemed to be “filed” for purposes
of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities
of that section, unless the Company specifically states that the information is to be considered “filed” under the Exchange
Act or specifically incorporates it by reference into a filing under the Securities Act of 1933, as amended, or the Exchange Act.
| Item 9.01 |
Financial Statements and Exhibits. |
(d) Exhibits.
SIGNATURES
Pursuant to the requirements of
the Securities Exchange Act of 1934, the Registrant has duly caused this report to be signed on its behalf by the undersigned hereunto
duly authorized.
Dated: March 11, 2024
| ZURA BIO LIMITED |
|
| |
|
| By: |
/s/ Kim Davis |
|
| |
Kim Davis |
|
| |
Chief Legal Officer |
|
Exhibit 99.1

| ©2024 Zura Bio Ltd.
Nasdaq Ticker: ZURA
Building the Next
Immunology Leader
March 2024 |

| ©2024 Zura Bio Ltd. 2
Forward Looking Statements Disclaimer
This communication includes “forward-looking statements” within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995.
Words such as “expect,” “estimate,” “project,” “budget,” “forecast,” “anticipate,” “intend,” “plan,” “may,” “will,” “could,” “should,” “believe,” “predict,” “potential,” “continue,”
“strategy,” “future,” “opportunity,” “would,” “seem,” “seek,” “outlook” and similar expressions are intended to identify such forward-looking statements. Forward-looking
statements are predictions, projections and other statements about future events that are based on current expectations and assumptions and, as a result, are
subject to risks and uncertainties that could cause the actual results to differ materially from the expected results. These statements are based on various
assumptions, whether or not identified in this communication. These forward-looking statements are provided for illustrative purposes only and are not intended to
serve as, and must not be relied on by an investor as, a guarantee, an assurance, a prediction or a definitive statement of fact or probability.
Actual events and circumstances are difficult or impossible to predict and will differ from assumptions. You should carefully consider the risks and uncertainties
described in the “Risk Factors” sections of Zura Bio’s recent filings with the SEC. These filings would identify and address other important risks and uncertainties that
could cause actual events and results to differ materially from those contained in the forward-looking statements. Many of these factors are outside Zura Bio’s control
and are difficult to predict. Many factors could cause actual future events to differ from the forward-looking statements in this communication, including but not
limited to: (1) the outcome of any legal proceedings that may be instituted against Zura Bio; (2) volatility in the price of Zura Bio’s securities; (3) the ability of Zura Bio to
successfully conduct research and development activities, grow and manage growth profitably, maintain relationships with customers and suppliers, and retain key
employees; (4) costs related to financing transactions and the ongoing costs relating to operating as a public company; (5) changes in the applicable laws or
regulations; (6) the possibility that Zura Bio may be adversely affected by other economic, business, and/or competitive factors; (7) the risk of downturns and a
changing regulatory landscape in the highly competitive industry in which Zura Bio operates; (8) the impact of the global COVID-19 pandemic; (9) the potential
inability of Zura Bio to raise additional capital needed to pursue its business objectives or to achieve efficiencies regarding other costs; (10) the enforceability of Zura
Bio’s intellectual property, including its patents, and the potential infringement on the intellectual property rights of others, cyber security risks or potential breaches
of data security; and (11) other risks and uncertainties described in the Registration Statement and such other documents filed by Zura Bio from time to time with
the SEC. These risks and uncertainties may be amplified by the COVID-19 pandemic or other unanticipated global disruption events, which may continue to cause
economic uncertainty. Zura Bio cautions that the foregoing list of factors is not exclusive or exhaustive and not to place undue reliance upon any forward-looking
statements, including projections, which speak only as of the date made. Zura Bio gives no assurance that it will achieve its expectations.
Zura Bio does not undertake or accept any obligation to publicly provide revisions or updates to any forward-looking statements, whether as a result of new
information, future developments or otherwise, or should circumstances change, except as otherwise required by securities and other applicable laws. |
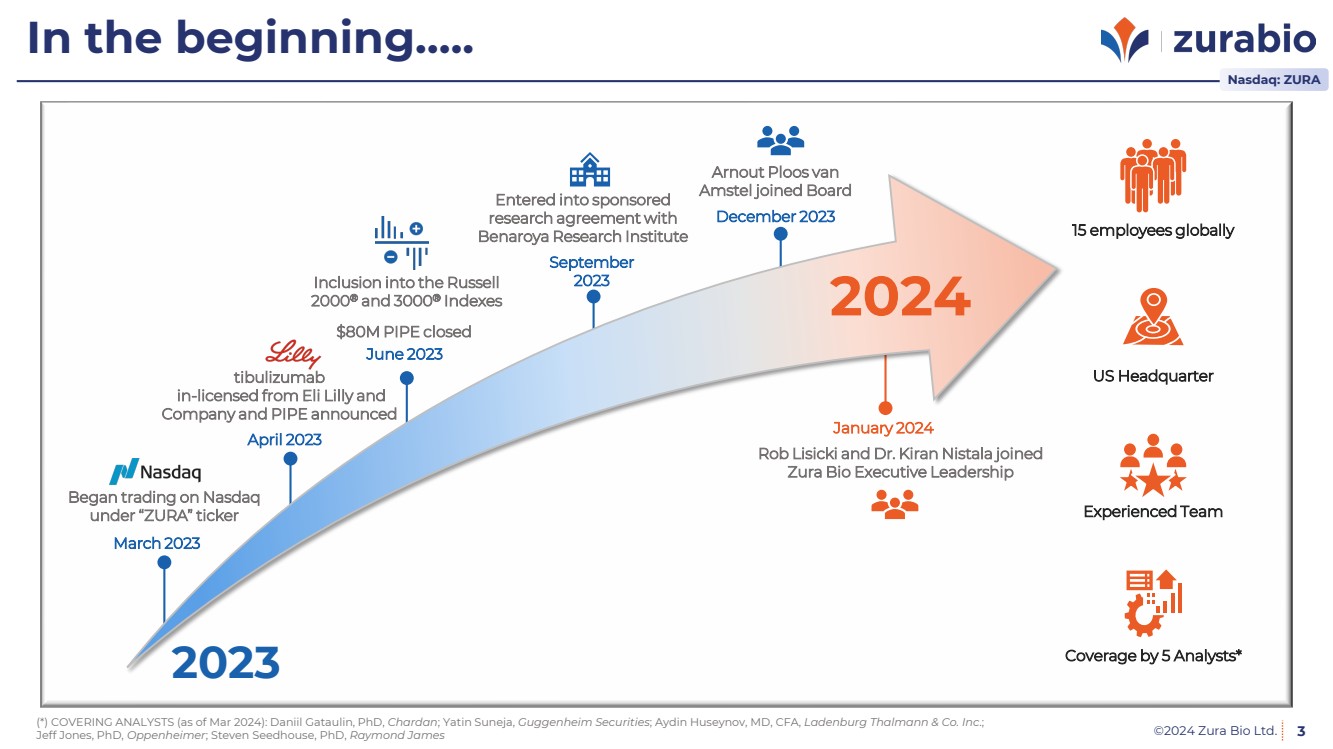
| ©2024 Zura Bio Ltd. 3
In the beginning…..
15 employees globally
Coverage by 5 Analysts*
US Headquarter
Experienced Team Began trading on Nasdaq
under “ZURA” ticker
tibulizumab
in-licensed from Eli Lilly and
Company and PIPE announced
$80M PIPE closed
Entered into sponsored
research agreement with
Benaroya Research Institute
Arnout Ploos van
Amstel joined Board
Rob Lisicki and Dr. Kiran Nistala joined
Zura Bio Executive Leadership
Inclusion into the Russell
2000® and 3000® Indexes
March 2023
2023
April 2023
June 2023
September
2023
December 2023
January 2024
2024
(*) COVERING ANALYSTS (as of Mar 2024): Daniil Gataulin, PhD, Chardan; Yatin Suneja, Guggenheim Securities; Aydin Huseynov, MD, CFA, Ladenburg Thalmann & Co. Inc.;
Jeff Jones, PhD, Oppenheimer; Steven Seedhouse, PhD, Raymond James
Nasdaq: ZURA |
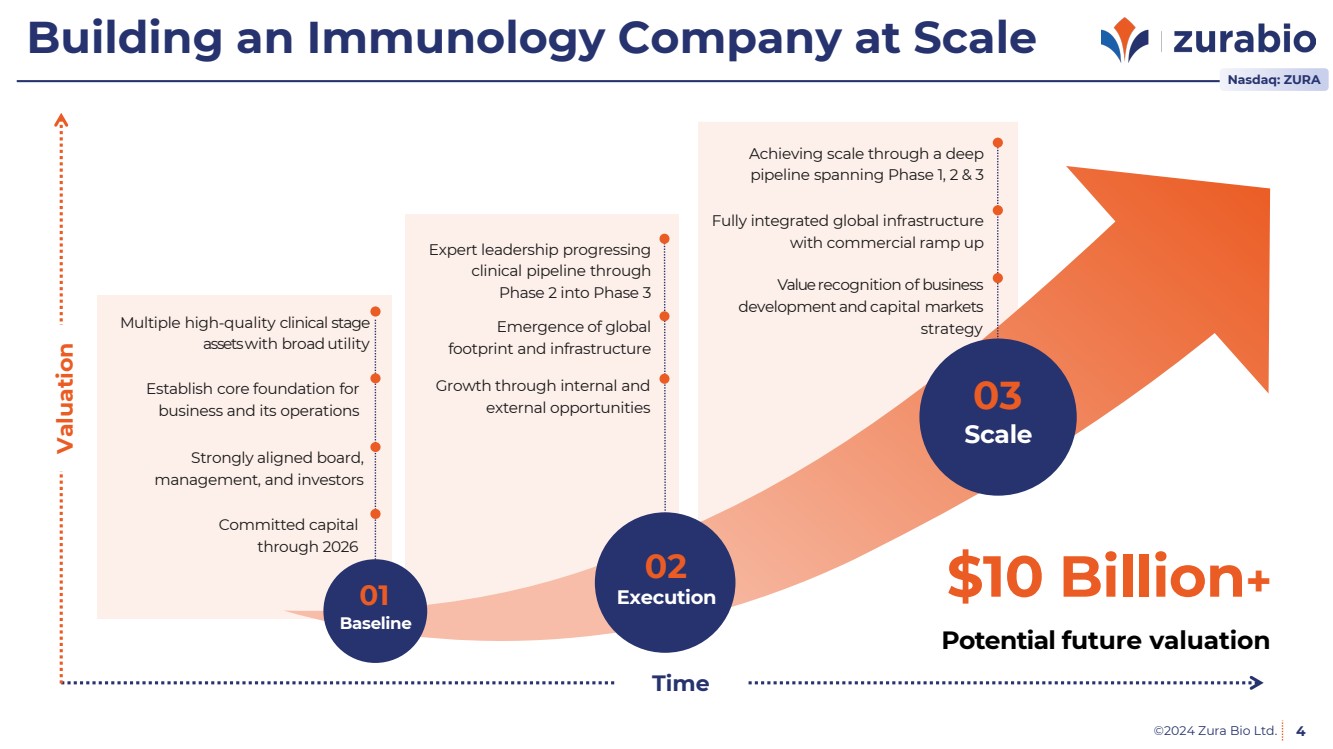
| ©2024 Zura Bio Ltd. 4
Building an Immunology Company at Scale
Establish core foundation for
business and its operations
Strongly aligned board,
management, and investors
Committed capital
through 2026
Multiple high-quality clinical stage
assetswith broadutility
Baseline
Execution $10 Billion+
Time
Valuation
Scale
01
02
03
Potential future valuation
Emergence of global
footprint and infrastructure
Growth through internal and
external opportunities
Expert leadership progressing
clinical pipeline through
Phase 2 into Phase 3
Fully integrated global infrastructure
with commercial ramp up
Valuerecognition of business
development and capital markets
strategy
Achieving scale through a deep
pipeline spanning Phase 1, 2 & 3
Nasdaq: ZURA |
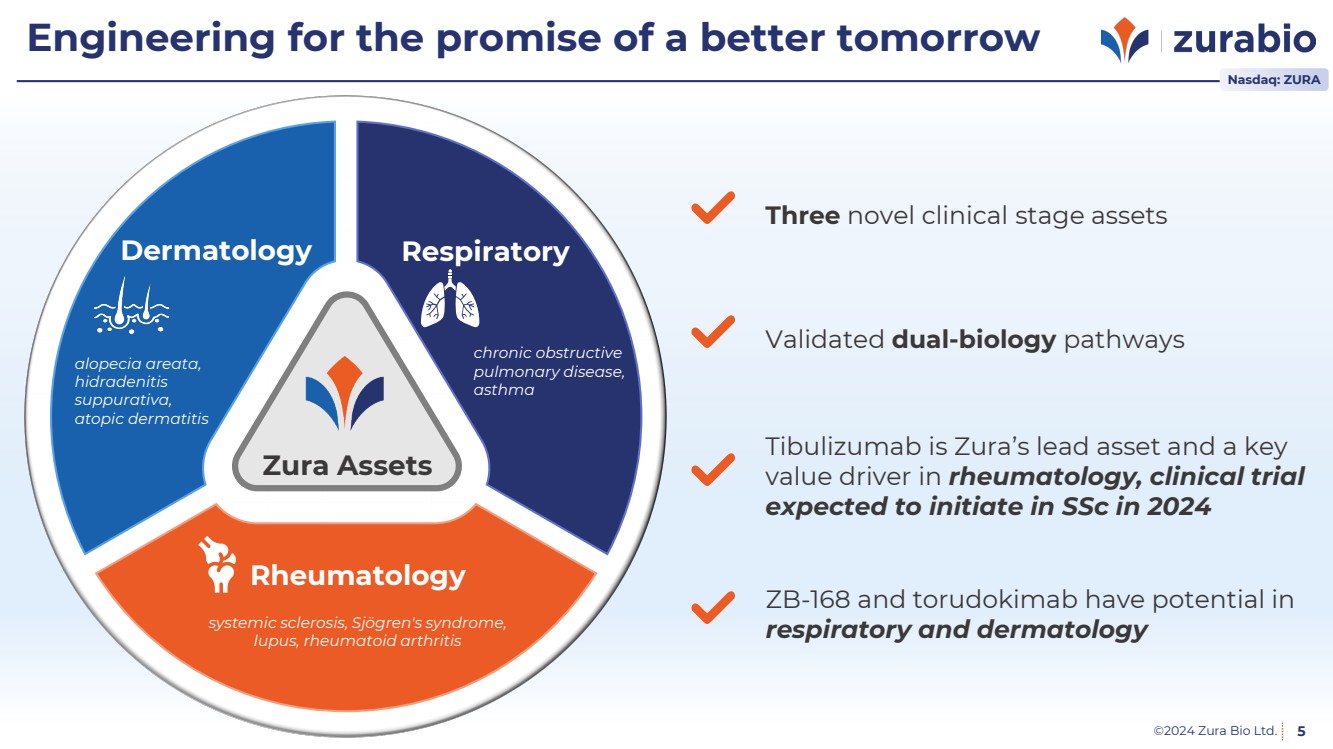
| ©2024 Zura Bio Ltd. 5
Engineering for the promise of a better tomorrow
Zura Assets
Rheumatology
systemic sclerosis, Sjögren's syndrome,
lupus, rheumatoid arthritis
Respiratory
chronic obstructive
pulmonary disease,
asthma
Dermatology
alopecia areata,
hidradenitis
suppurativa,
atopic dermatitis
Validated dual-biology pathways Created by IYIKON
from the Noun Project
Three novel clinical stage assets Created by IYIKON
from the Noun Project
Tibulizumab is Zura’s lead asset and a key
value driver in rheumatology, clinical trial
expected to initiate in SSc in 2024 Created by IYIKON
from the Noun Project
ZB-168 and torudokimab have potential in
Created by IYIKON
from the Noun Project respiratory and dermatology
Nasdaq: ZURA |
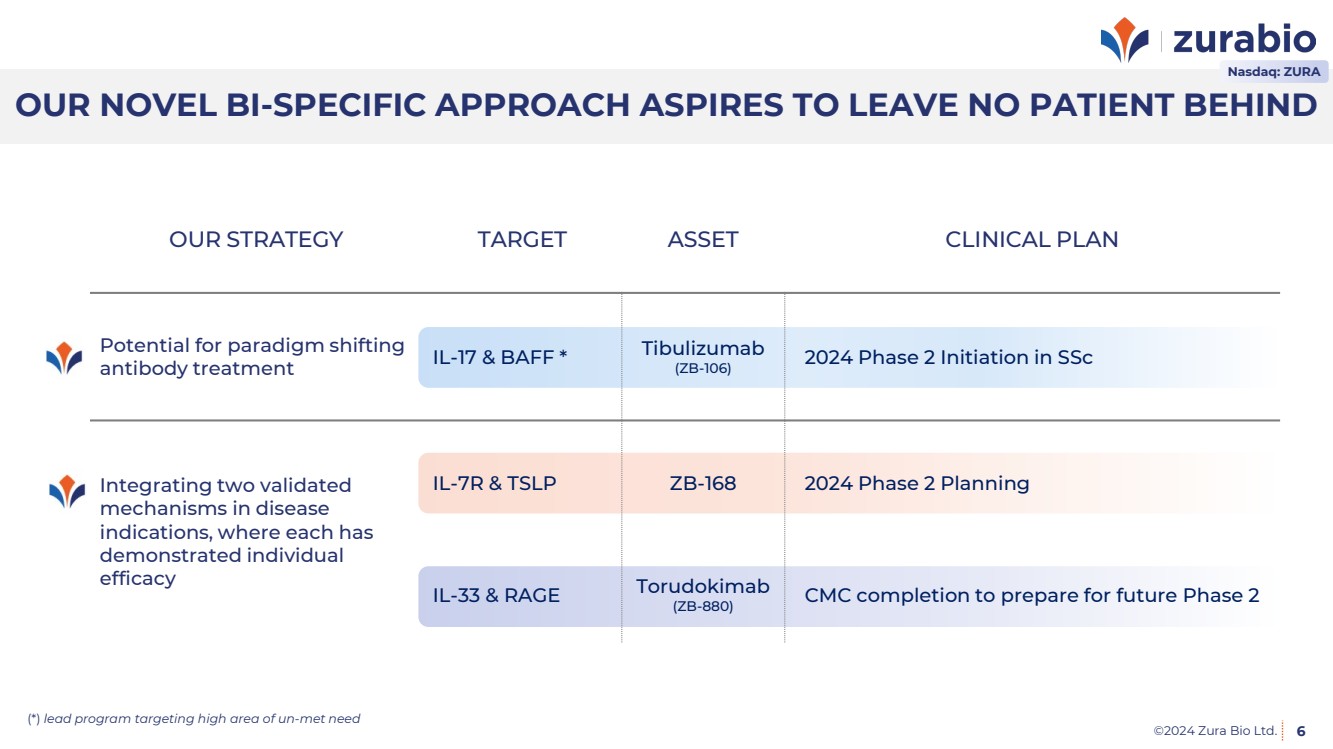
| ©2024 Zura Bio Ltd.
OUR NOVEL BI-SPECIFIC APPROACH ASPIRES TO LEAVE NO PATIENT BEHIND
6
OUR STRATEGY TARGET ASSET CLINICAL PLAN
Potential for paradigm shifting
antibody treatment IL-17 & BAFF * Tibulizumab
(ZB-106) 2024 Phase 2 Initiation in SSc
Integrating two validated
mechanisms in disease
indications, where each has
demonstrated individual
efficacy
IL-7R & TSLP ZB-168 2024 Phase 2 Planning
IL-33 & RAGE Torudokimab
(ZB-880) CMC completion to prepare for future Phase 2
(*) lead program targeting high area of un-met need
Nasdaq: ZURA |
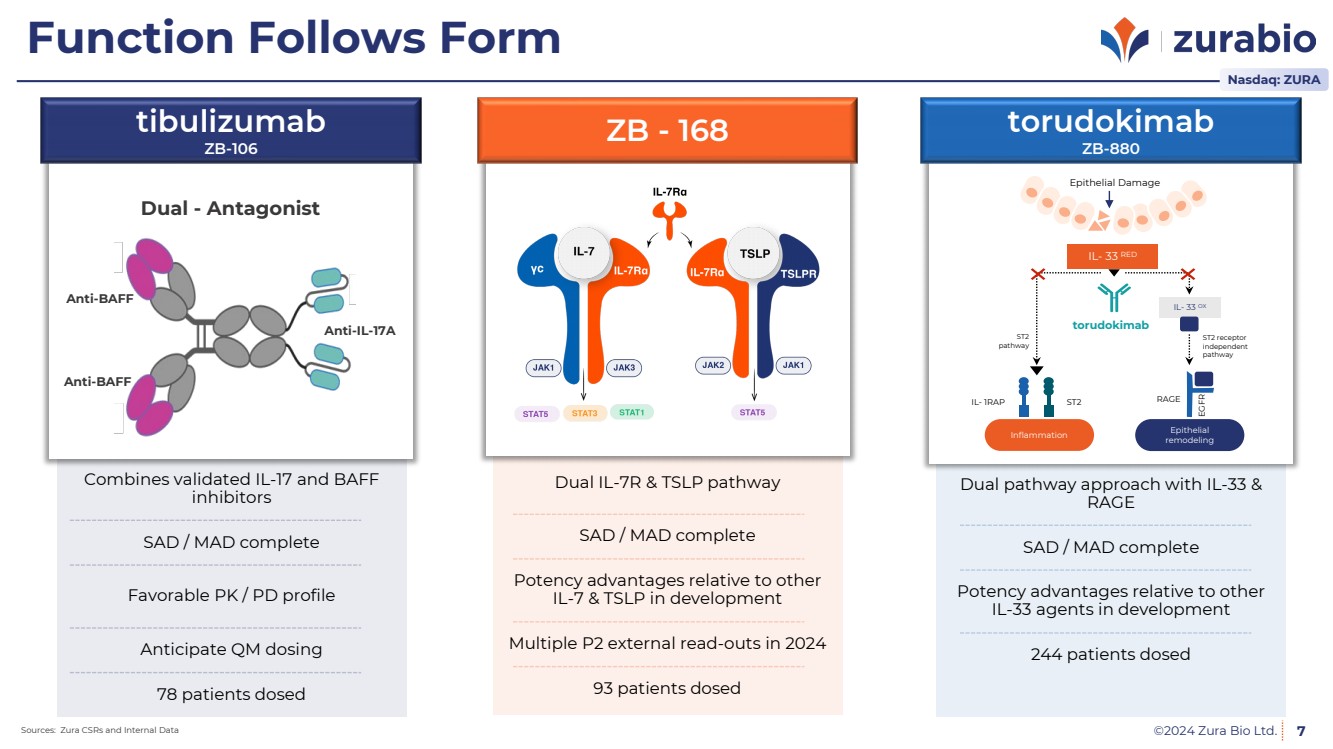
| ©2024 Zura Bio Ltd. 7
Function Follows Form
Nasdaq: ZURA
Sources: Zura CSRs and Internal Data
93 patients dosed
Multiple P2 external read-outs in 2024
Dual IL-7R & TSLP pathway
SAD / MAD complete
Potency advantages relative to other
IL-7 & TSLP in development
ZB - 168
244 patients dosed
Dual pathway approach with IL-33 &
RAGE
SAD / MAD complete
Potency advantages relative to other
IL-33 agents in development
7
IL- 33 RED
IL- 33 OX
RAGE
EGFR
ST2 receptor
independent
pathway
Epithelial
remodeling
IL- 1RAP ST2
Inflammation
ST2
pathway
Epithelial Damage
torudokimab
torudokimab
ZB-880
78 patients dosed
Anticipate QM dosing
Combines validated IL-17 and BAFF
inhibitors
SAD / MAD complete
Favorable PK / PD profile
z
Dual - Antagonist
tibulizumab
ZB-106
Anti-BAFF
Anti-IL-17A
Anti-BAFF |

| ©2024 Zura Bio Ltd.
Potential First-in-Class, Dual
Antagonist Combining
tabalumab and TALTZ®
systemic sclerosis (SSc)
tibulizumab
ZB-106
Anti-BAFF x IL-17 |
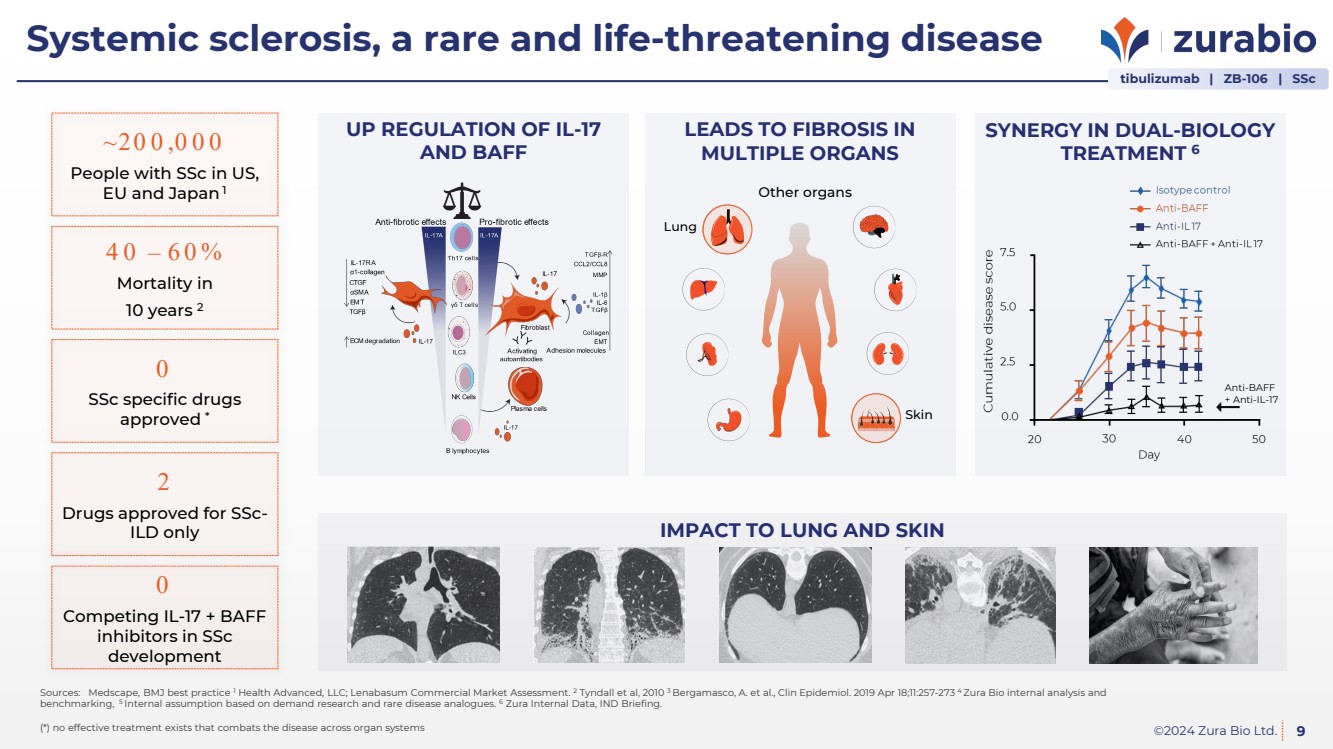
| ©2024 Zura Bio Ltd.
Systemic sclerosis, a rare and life-threatening disease
9
Sources: Medscape, BMJ best practice 1 Health Advanced, LLC; Lenabasum Commercial Market Assessment. 2 Tyndall et al, 2010 3 Bergamasco, A. et al., Clin Epidemiol. 2019 Apr 18;11:257-273 4 Zura Bio internal analysis and
benchmarking, 5 Internal assumption based on demand research and rare disease analogues. 6 Zura Internal Data, IND Briefing.
(*) no effective treatment exists that combats the disease across organ systems
~20 0 ,0 0 0
People with SSc in US,
EU and Japan1
40 – 60%
Mortality in
10 years 2
0
SSc specific drugs
approved *
2
Drugs approved for SSc-ILD only
0
Competing IL-17 + BAFF
inhibitors in SSc
development
LEADS TO FIBROSIS IN
MULTIPLE ORGANS
Lung
Other organs
Skin
UP REGULATION OF IL-17
AND BAFF
SYNERGY IN DUAL-BIOLOGY
TREATMENT 6
IMPACT TO LUNG AND SKIN
Anti-BAFF
+ Anti-IL-17
tibulizumab | ZB-106 | SSc |
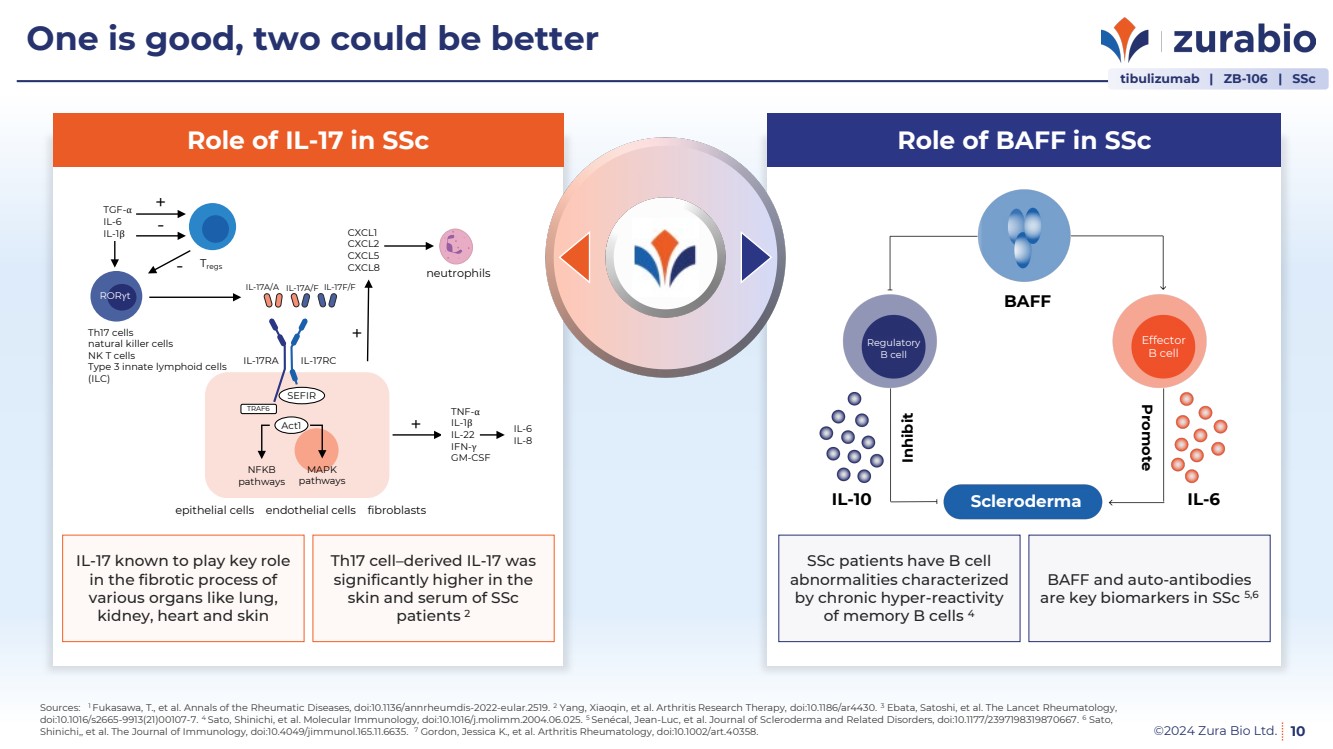
| ©2024 Zura Bio Ltd. 10
One is good, two could be better
Role of BAFF in SSc
BAFF and auto-antibodies
are key biomarkers in SSc 5,6
SSc patients have B cell
abnormalities characterized
by chronic hyper-reactivity
of memory B cells 4
BAFF
IL-10 IL-6 Inhibit Promote
Effector
B cell
Regulatory
B cell
Scleroderma
Sources: 1 Fukasawa, T., et al. Annals of the Rheumatic Diseases, doi:10.1136/annrheumdis-2022-eular.2519. 2 Yang, Xiaoqin, et al. Arthritis Research Therapy, doi:10.1186/ar4430. 3 Ebata, Satoshi, et al. The Lancet Rheumatology,
doi:10.1016/s2665-9913(21)00107-7. 4 Sato, Shinichi, et al. Molecular Immunology, doi:10.1016/j.molimm.2004.06.025. 5 Senécal, Jean-Luc, et al. Journal of Scleroderma and Related Disorders, doi:10.1177/2397198319870667. 6 Sato,
Shinichi,, et al. The Journal of Immunology, doi:10.4049/jimmunol.165.11.6635. 7 Gordon, Jessica K., et al. Arthritis Rheumatology, doi:10.1002/art.40358.
Role of IL-17 in SSc
Th17 cell–derived IL-17 was
significantly higher in the
skin and serum of SSc
patients 2
IL-17 known to play key role
in the fibrotic process of
various organs like lung,
kidney, heart and skin
+
+
epithelial cells endothelial cells fibroblasts
MAPK
pathways
NFKB
pathways
CXCL1
CXCL2
CXCL5
CXCL8 neutrophils
IL-17A/A IL-17A/F IL-17F/F
IL-17RA IL-17RC
- Tregs
TNF-α
IL-1β
IL-22
IFN-γ
GM-CSF
IL-6
IL-8
TGF-α
IL-6
IL-1β
+
-
Th17 cells
natural killer cells
NK T cells
Type 3 innate lymphoid cells
(ILC)
RORγt
SEFIR
Act1
TRAF6
tibulizumab | ZB-106 | SSc |
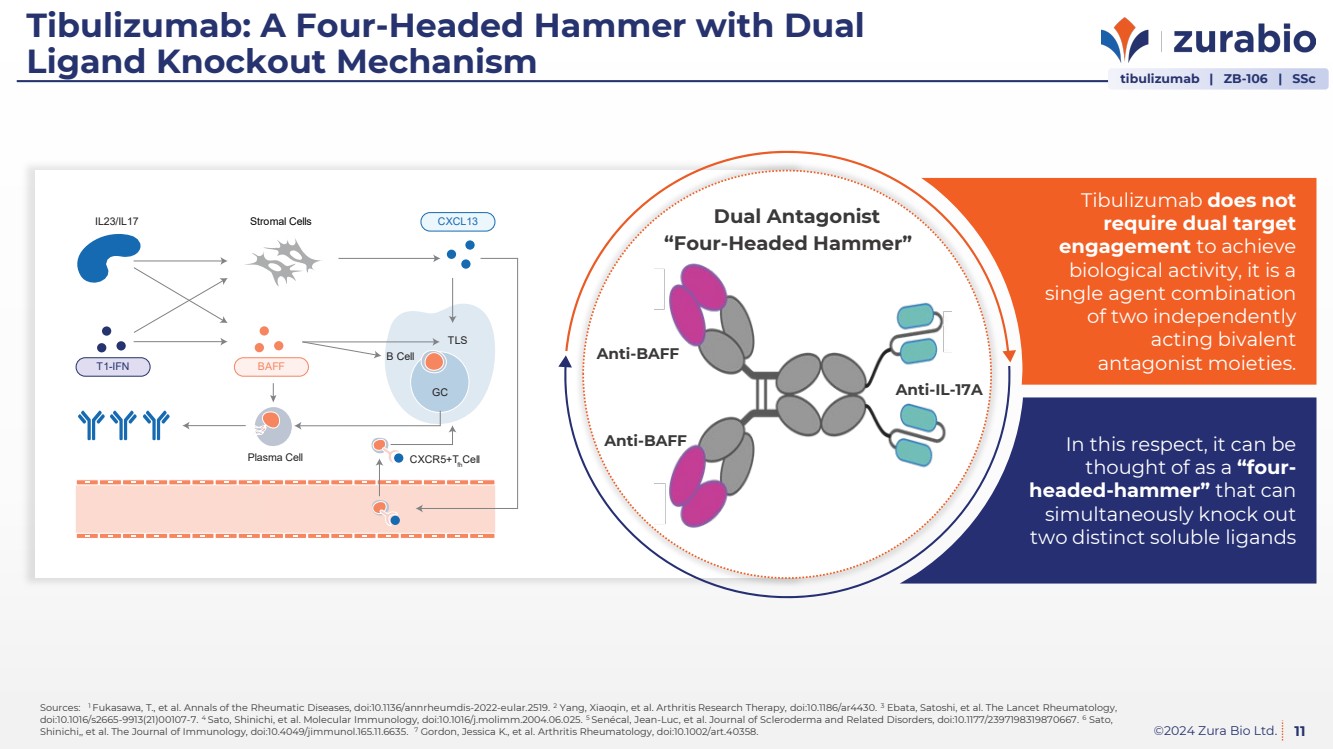
| ©2024 Zura Bio Ltd.
Tibulizumab: A Four-Headed Hammer with Dual
Ligand Knockout Mechanism
11
Sources: 1 Fukasawa, T., et al. Annals of the Rheumatic Diseases, doi:10.1136/annrheumdis-2022-eular.2519. 2 Yang, Xiaoqin, et al. Arthritis Research Therapy, doi:10.1186/ar4430. 3 Ebata, Satoshi, et al. The Lancet Rheumatology,
doi:10.1016/s2665-9913(21)00107-7. 4 Sato, Shinichi, et al. Molecular Immunology, doi:10.1016/j.molimm.2004.06.025. 5 Senécal, Jean-Luc, et al. Journal of Scleroderma and Related Disorders, doi:10.1177/2397198319870667. 6 Sato,
Shinichi,, et al. The Journal of Immunology, doi:10.4049/jimmunol.165.11.6635. 7 Gordon, Jessica K., et al. Arthritis Rheumatology, doi:10.1002/art.40358.
GC
TLS
B Cell
CXCL13
T1-IFN BAFF
Stromal Cells
Plasma Cell
IL23/IL17
CXCR5+TfhCell
In this respect, it can be
thought of as a “four-headed-hammer” that can
simultaneously knock out
two distinct soluble ligands
Tibulizumab does not
require dual target
engagement to achieve
biological activity, it is a
single agent combination
of two independently
acting bivalent
antagonist moieties. Anti-BAFF
Anti-IL-17A
Anti-BAFF
“Four-Headed Hammer”
Dual Antagonist
tibulizumab | ZB-106 | SSc |
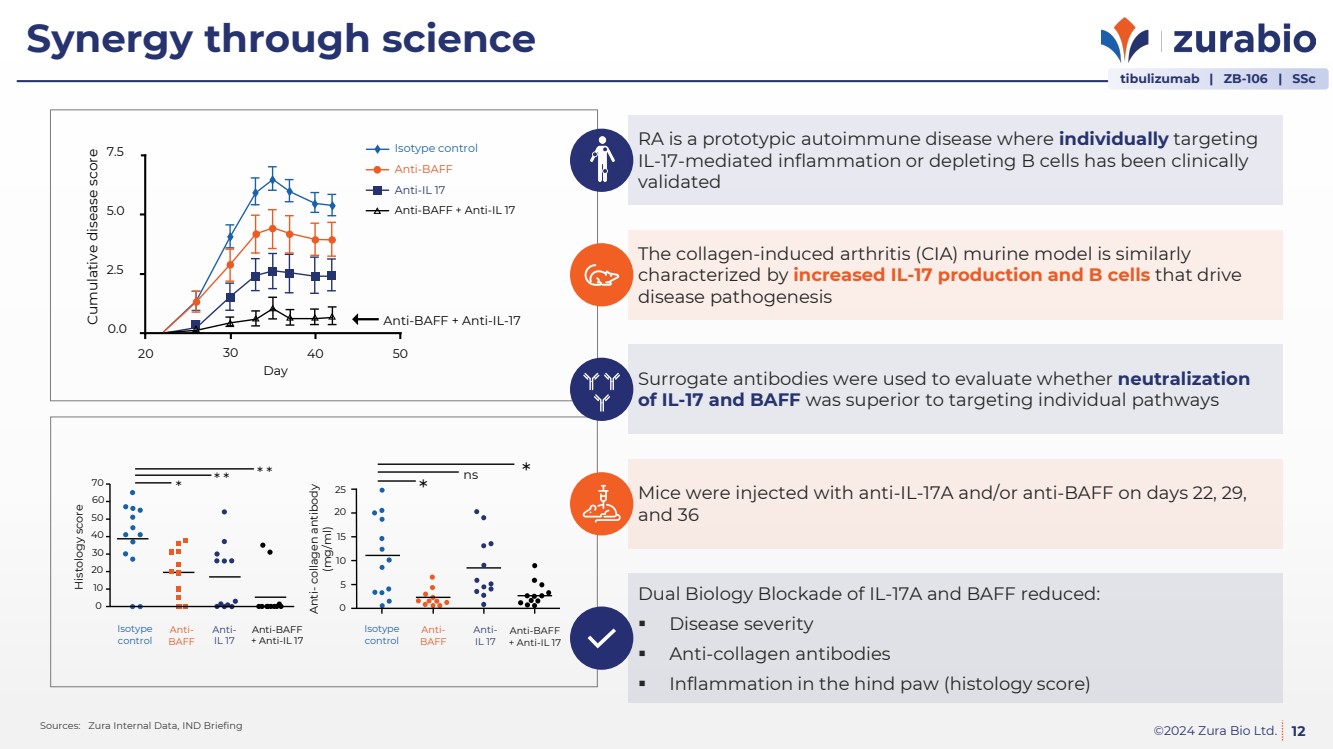
| ©2024 Zura Bio Ltd.
Synergy through science
12 Sources: Zura Internal Data, IND Briefing
Isotype control
Anti-BAFF
Anti-IL 17
Anti-BAFF + Anti-IL 17
Anti-BAFF + Anti-IL-17
7.5
5.0
2.5
0.0
20 30 40 50
Day
Cumulative disease score
Anti-BAFF
+ Anti-IL 17
Histology score
70
60
* * * * *
Anti-BAFF
+ Anti-IL 17
Isotype
control
Anti-BAFF
Anti-IL 17
50
40
30
20
10
0
*
25 *
20
15
10
5
0
Isotype
control
Anti-BAFF
Anti-IL 17
ns
Anti- collagen antibody
(mg/ml)
RA is a prototypic autoimmune disease where individually targeting
IL-17-mediated inflammation or depleting B cells has been clinically
validated
The collagen-induced arthritis (CIA) murine model is similarly
characterized by increased IL-17 production and B cells that drive
disease pathogenesis
Surrogate antibodies were used to evaluate whether neutralization
of IL-17 and BAFF was superior to targeting individual pathways
Dual Biology Blockade of IL-17A and BAFF reduced:
Disease severity
Anti-collagen antibodies
Inflammation in the hind paw (histology score)
Mice were injected with anti-IL-17A and/or anti-BAFF on days 22, 29,
and 36
tibulizumab | ZB-106 | SSc |
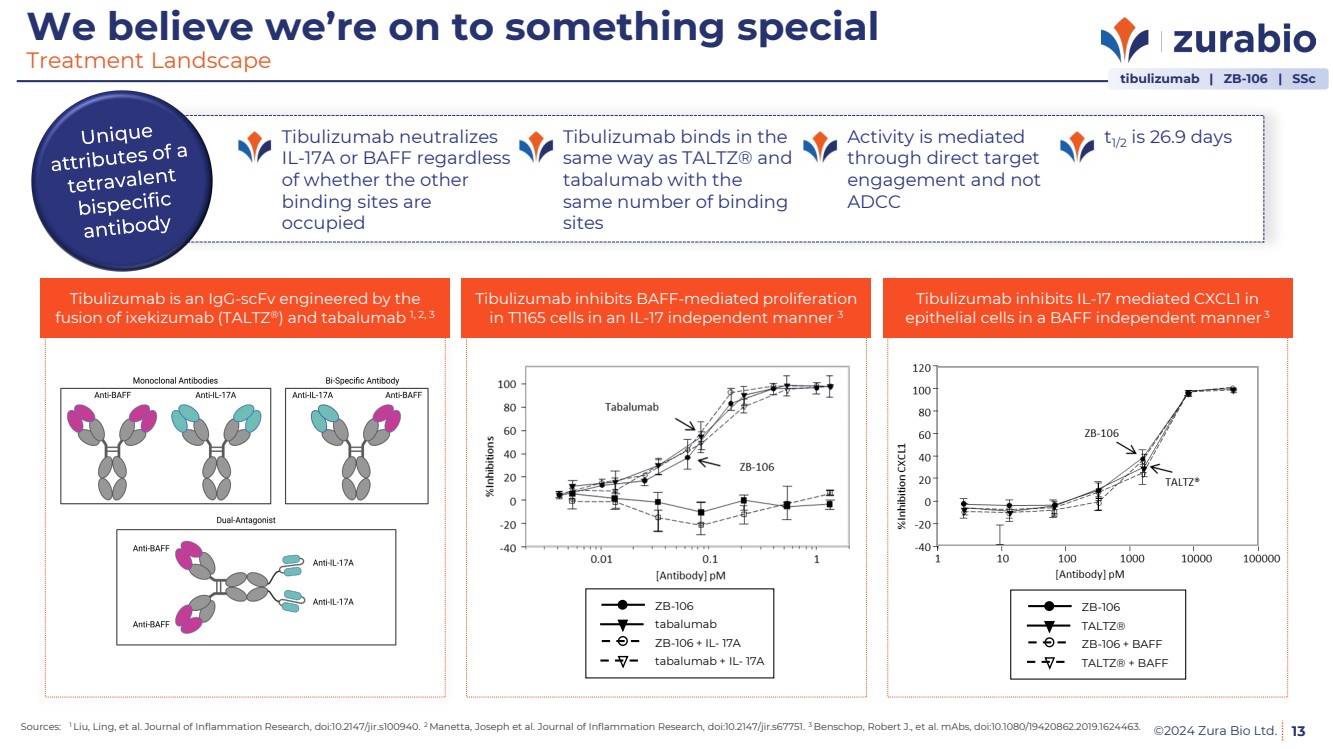
| ©2024 Zura Bio Ltd. 13
We believe we’re on to something special
Treatment Landscape
Tibulizumab neutralizes
IL-17A or BAFF regardless
of whether the other
binding sites are
occupied
Tibulizumab binds in the
same way as TALTZ® and
tabalumab with the
same number of binding
sites
Activity is mediated
through direct target
engagement and not
ADCC
t1/2 is 26.9 days
tibulizumab | ZB-106 | SSc
Tibulizumab is an IgG-scFv engineered by the
fusion of ixekizumab (TALTZ®) and tabalumab 1, 2, 3
Tibulizumab inhibits BAFF-mediated proliferation
in T1165 cells in an IL-17 independent manner 3
Tibulizumab inhibits IL-17 mediated CXCL1 in
epithelial cells in a BAFF independent manner 3
tabalumab
tabalumab + IL- 17A
ZB-106
ZB-106 + IL- 17A
TALTZ®
TALTZ® + BAFF
ZB-106
ZB-106 + BAFF
Sources: 1 Liu, Ling, et al. Journal of Inflammation Research, doi:10.2147/jir.s100940. 2 Manetta, Joseph et al. Journal of Inflammation Research, doi:10.2147/jir.s67751. 3 Benschop, Robert J., et al. mAbs, doi:10.1080/19420862.2019.1624463. |
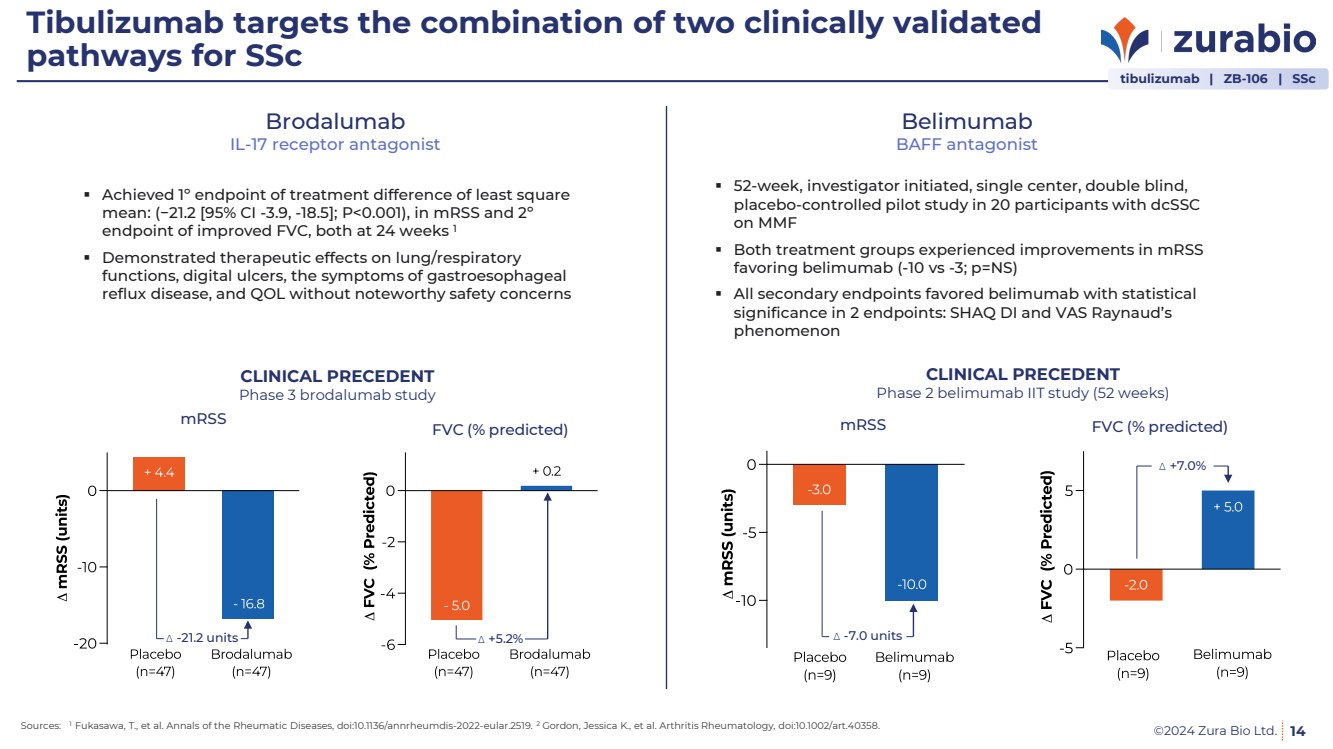
| ©2024 Zura Bio Ltd.
Tibulizumab targets the combination of two clinically validated
pathways for SSc
14
Brodalumab
IL-17 receptor antagonist
Achieved 1º endpoint of treatment difference of least square
mean: (−21.2 [95% CI -3.9, -18.5]; P<0.001), in mRSS and 2º
endpoint of improved FVC, both at 24 weeks 1
Demonstrated therapeutic effects on lung/respiratory
functions, digital ulcers, the symptoms of gastroesophageal
reflux disease, and QOL without noteworthy safety concerns
Belimumab
BAFF antagonist
52-week, investigator initiated, single center, double blind,
placebo-controlled pilot study in 20 participants with dcSSC
on MMF
Both treatment groups experienced improvements in mRSS
favoring belimumab (-10 vs -3; p=NS)
All secondary endpoints favored belimumab with statistical
significance in 2 endpoints: SHAQ DI and VAS Raynaud’s
phenomenon
Sources: 1 Fukasawa, T., et al. Annals of the Rheumatic Diseases, doi:10.1136/annrheumdis-2022-eular.2519. 2 Gordon, Jessica K., et al. Arthritis Rheumatology, doi:10.1002/art.40358.
Δ -21.2 units Δ +5.2%
CLINICAL PRECEDENT
Phase 3 brodalumab study
mRSS FVC (% predicted)
CLINICAL PRECEDENT
Phase 2 belimumab IIT study (52 weeks)
mRSS FVC (% predicted)
Δ -7.0 units
Δ +7.0%
tibulizumab | ZB-106 | SSc |
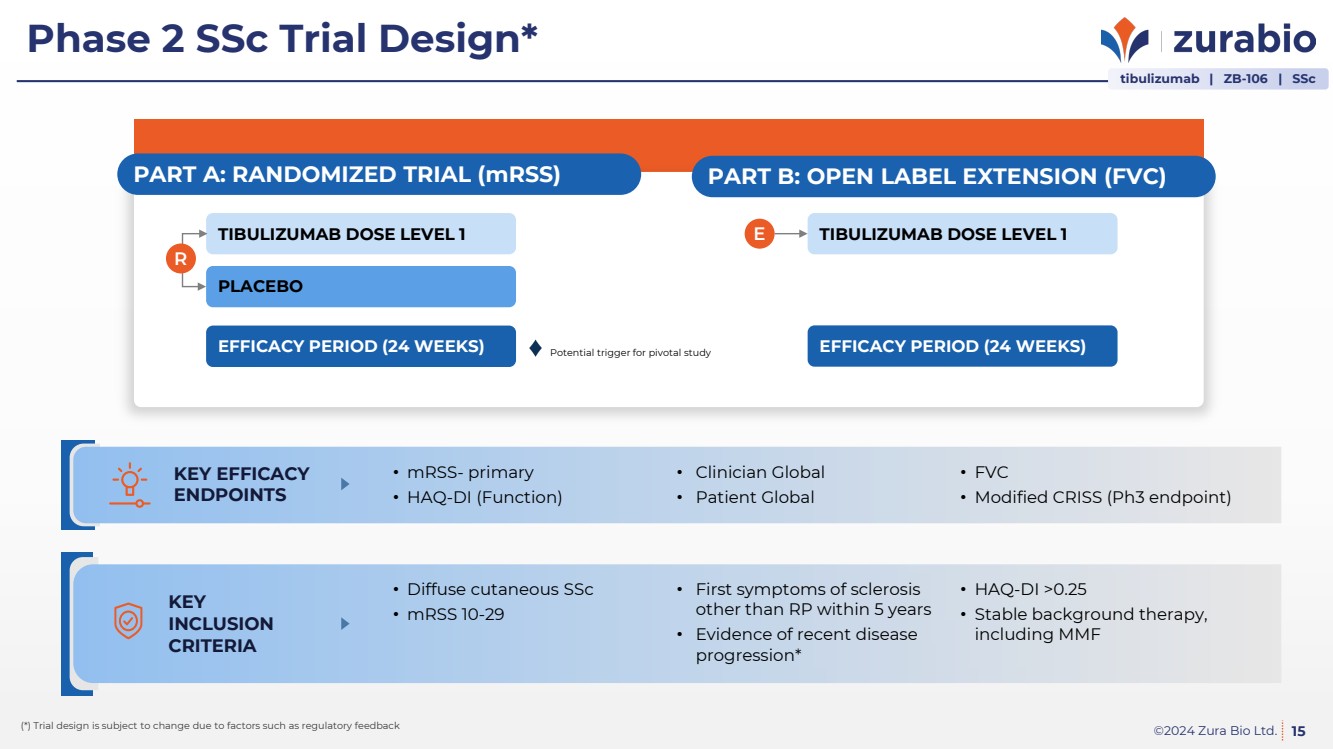
| ©2024 Zura Bio Ltd.
Phase 2 SSc Trial Design*
15 (*) Trial design is subject to change due to factors such as regulatory feedback
PART A: RANDOMIZED TRIAL (mRSS)
EFFICACY PERIOD (24 WEEKS)
PLACEBO
TIBULIZUMAB DOSE LEVEL 1
R
PART B: OPEN LABEL EXTENSION (FVC)
EFFICACY PERIOD (24 WEEKS)
E TIBULIZUMAB DOSE LEVEL 1
KEY EFFICACY
ENDPOINTS
• mRSS- primary
• HAQ-DI (Function)
• Clinician Global
• Patient Global
• FVC
• Modified CRISS (Ph3 endpoint)
KEY
INCLUSION
CRITERIA
• Diffuse cutaneous SSc
• mRSS 10-29
• First symptoms of sclerosis
other than RP within 5 years
• Evidence of recent disease
progression*
• HAQ-DI >0.25
• Stable background therapy,
including MMF
Potential trigger for pivotal study
tibulizumab | ZB-106 | SSc |
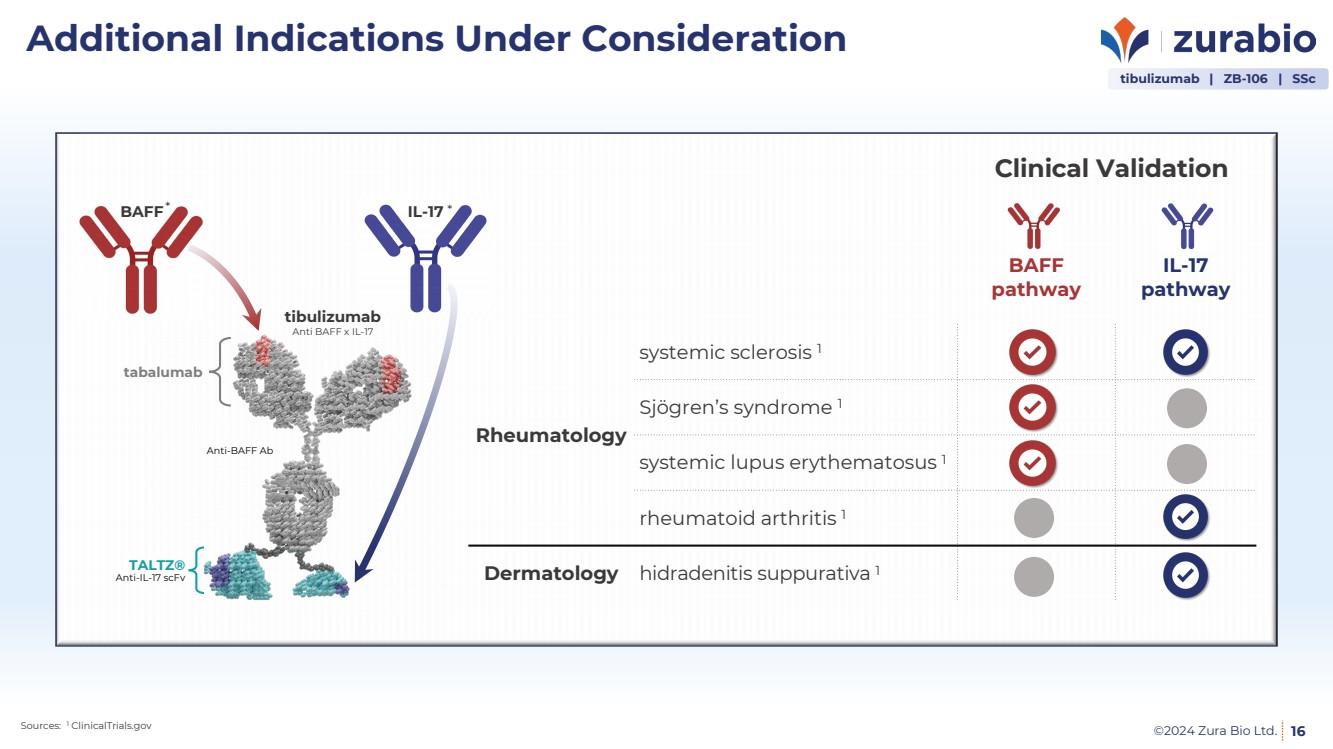
| ©2024 Zura Bio Ltd.
Additional Indications Under Consideration
16
tibulizumab | ZB-106 | SSc
BAFF
pathway
IL-17
pathway
Rheumatology
systemic sclerosis 1
Sjögren’s syndrome 1
systemic lupus erythematosus 1
rheumatoid arthritis 1
Dermatology hidradenitis suppurativa 1
Sources: 1 ClinicalTrials.gov
BAFF IL-17
Anti-IL-17 scFv
TALTZ®
Anti-BAFF Ab
tabalumab
tibulizumab
Anti BAFF x IL-17
* *
Clinical Validation |

| ©2024 Zura Bio Ltd.
Product Candidates
ZB-168 &
torudokimab |
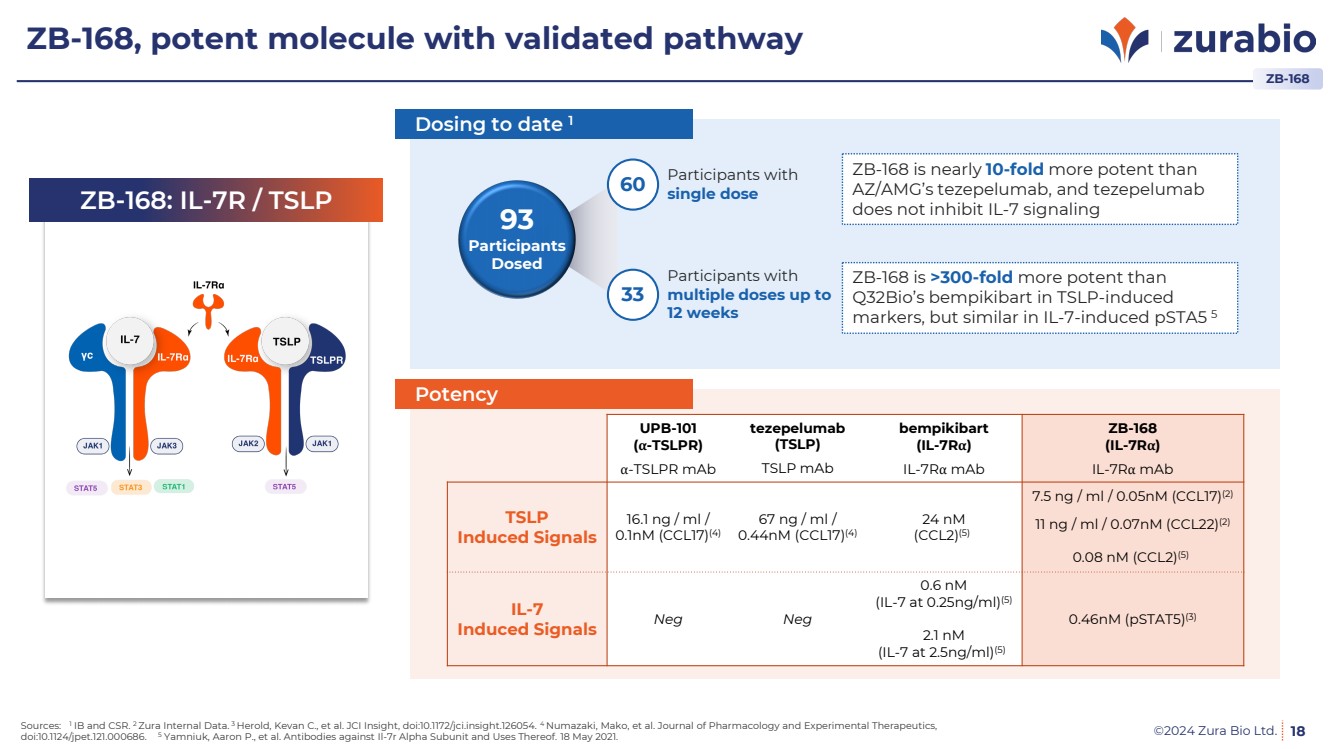
| ©2024 Zura Bio Ltd.
ZB-168, potent molecule with validated pathway
18
UPB-101
(α-TSLPR)
tezepelumab
(TSLP)
bempikibart
(IL-7Rα)
ZB-168
(IL-7Rα)
α-TSLPR mAb TSLP mAb IL-7Rα mAb IL-7Rα mAb
TSLP
Induced Signals
16.1 ng / ml /
0.1nM (CCL17)(4)
67 ng / ml /
0.44nM (CCL17)(4)
24 nM
(CCL2)(5)
7.5 ng / ml / 0.05nM (CCL17)(2)
11 ng / ml / 0.07nM (CCL22)(2)
0.08 nM (CCL2)(5)
IL-7
Induced Signals Neg Neg
0.6 nM
(IL-7 at 0.25ng/ml)(5)
2.1 nM
(IL-7 at 2.5ng/ml)(5)
0.46nM (pSTAT5)(3)
ZB-168: IL-7R / TSLP 93
Participants
Dosed
Participants with
single dose 60
Participants with
multiple doses up to
12 weeks
33
ZB-168 is nearly 10-fold more potent than
AZ/AMG’s tezepelumab, and tezepelumab
does not inhibit IL-7 signaling
ZB-168 is >300-fold more potent than
Q32Bio’s bempikibart in TSLP-induced
markers, but similar in IL-7-induced pSTA5 5
Dosing to date 1
Potency
Sources: 1 IB and CSR. 2 Zura Internal Data. 3 Herold, Kevan C., et al. JCI Insight, doi:10.1172/jci.insight.126054. 4 Numazaki, Mako, et al. Journal of Pharmacology and Experimental Therapeutics,
doi:10.1124/jpet.121.000686. 5 Yamniuk, Aaron P., et al. Antibodies against Il-7r Alpha Subunit and Uses Thereof. 18 May 2021.
ZB-168 |
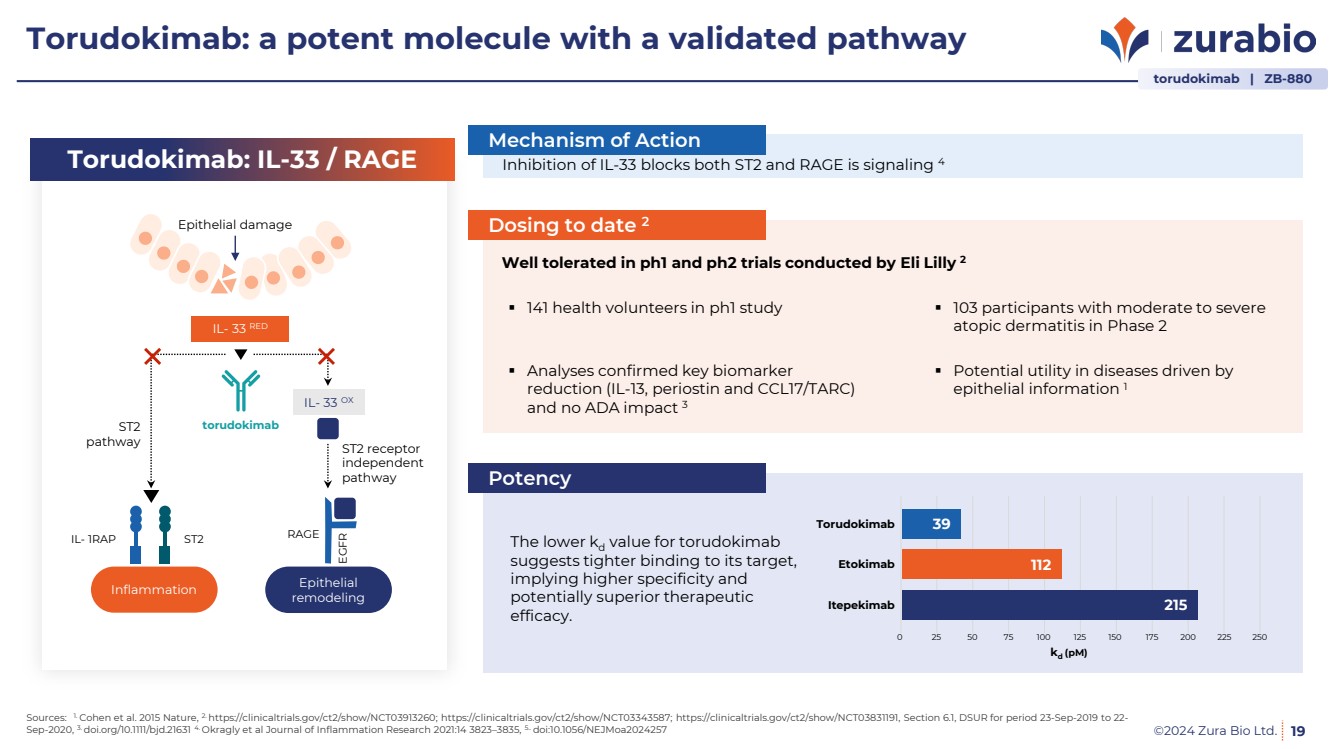
| ©2024 Zura Bio Ltd.
Torudokimab: a potent molecule with a validated pathway
19
Inhibition of IL-33 blocks both ST2 and RAGE is signaling 4
Well tolerated in ph1 and ph2 trials conducted by Eli Lilly 2
141 health volunteers in ph1 study
Analyses confirmed key biomarker
reduction (IL-13, periostin and CCL17/TARC)
and no ADA impact 3
Potential utility in diseases driven by
epithelial information 1
103 participants with moderate to severe
atopic dermatitis in Phase 2
The lower kd value for torudokimab
suggests tighter binding to its target,
implying higher specificity and
potentially superior therapeutic
efficacy.
0 25 50 75 100 125 150 175 200 225 250
Torudokimab
Etokimab
Itepekimab
39
112
215
kd (pM)
IL- 33 RED
IL- 33 OX
RAGE
EGFR
ST2 receptor
independent
pathway
Epithelial
remodeling
IL- 1RAP ST2
Inflammation
ST2
pathway
Epithelial damage
torudokimab
Mechanism of Action
Dosing to date 2
Potency
Torudokimab: IL-33 / RAGE
torudokimab | ZB-880
Sources: 1. Cohen et al. 2015 Nature, 2. https://clinicaltrials.gov/ct2/show/NCT03913260; https://clinicaltrials.gov/ct2/show/NCT03343587; https://clinicaltrials.gov/ct2/show/NCT03831191, Section 6.1, DSUR for period 23-Sep-2019 to 22-
Sep-2020, 3. doi.org/10.1111/bjd.21631 4. Okragly et al Journal of Inflammation Research 2021:14 3823–3835, 5.. doi:10.1056/NEJMoa2024257 |
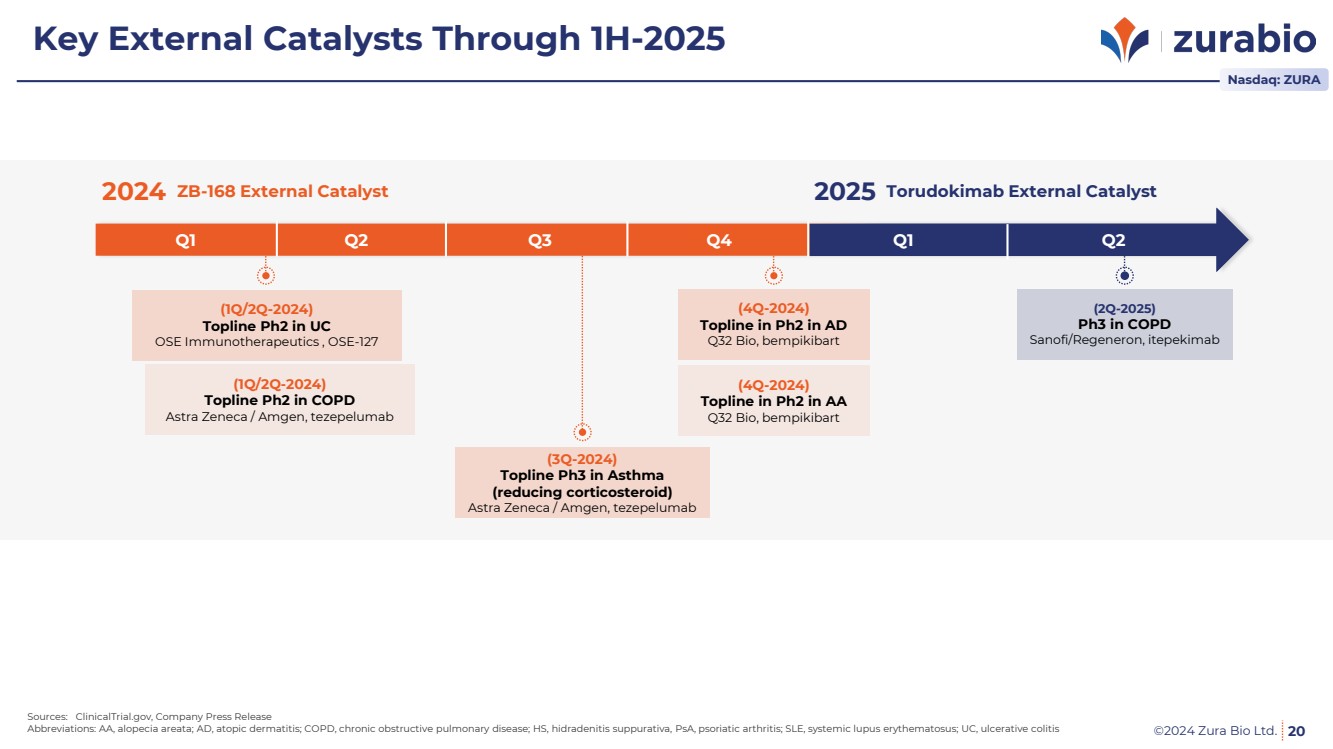
| ©2024 Zura Bio Ltd. 20
Sources: ClinicalTrial.gov, Company Press Release
Abbreviations: AA, alopecia areata; AD, atopic dermatitis; COPD, chronic obstructive pulmonary disease; HS, hidradenitis suppurativa, PsA, psoriatic arthritis; SLE, systemic lupus erythematosus; UC, ulcerative colitis
Nasdaq: ZURA
Key External Catalysts Through 1H-2025
ZB-168 External Catalyst
(1Q/2Q-2024)
Topline Ph2 in UC
OSE Immunotherapeutics , OSE-127
(1Q/2Q-2024)
Topline Ph2 in COPD
Astra Zeneca / Amgen, tezepelumab
2024 2025
(3Q-2024)
Topline Ph3 in Asthma
(reducing corticosteroid)
Astra Zeneca / Amgen, tezepelumab
(4Q-2024)
Topline in Ph2 in AD
Q32 Bio, bempikibart
(4Q-2024)
Topline in Ph2 in AA
Q32 Bio, bempikibart
Torudokimab External Catalyst
Q1 Q2 Q3 Q4 Q1 Q2
(2Q-2025)
Ph3 in COPD
Sanofi/Regeneron, itepekimab |

| Nasdaq Ticker: ZURA
2024 Key Objectives:
On time clinical trial execution
Build leadership team with specific expertise
Translational excellence & validating external
clinical readouts |

| ©2024 Zura Bio Ltd. 22
Experienced management team with proven ability to
successfully execute and build a leading market position
Board of Directors
Executive Team
Amit Munshi Arnout Ploos van Amstel Jennifer Jarrett Neil Graham, M.D. Parvinder Thiara Sandeep Kulkarni, M.D. Someit Sidhu, M.D. Steve Schoch
Chairman Independent Director Independent Director Independent Director Independent Director Independent Director Director Independent Director
Mike Howell Ph.D.
Chief Scientific Officer and
Head of Translational Medicine
Kim Davis
Chief Legal Officer
Kiran Nistala M.D., Ph.D.
Chief Medical Officer and
Head of Development
Gary Whale Ph.D.
Chief Technology Officer
Someit Sidhu M.D.
Founder, Chief Executive Officer and
Director
Verender Badial
Chief Financial Officer
Robert Lisicki
President and
Chief Operating Officer
Nasdaq: ZURA |
v3.24.0.1
Cover
|
Mar. 11, 2024 |
| Document Information [Line Items] |
|
| Document Type |
8-K
|
| Amendment Flag |
false
|
| Document Period End Date |
Mar. 11, 2024
|
| Entity File Number |
001-40598
|
| Entity Registrant Name |
Zura
Bio Limited
|
| Entity Central Index Key |
0001855644
|
| Entity Tax Identification Number |
98-1725736
|
| Entity Incorporation, State or Country Code |
E9
|
| Entity Address, Address Line One |
1489 W. Warm Springs Rd.
|
| Entity Address, Address Line Two |
#110
|
| Entity Address, City or Town |
Henderson
|
| Entity Address, State or Province |
NV
|
| Entity Address, Postal Zip Code |
89014
|
| City Area Code |
702
|
| Local Phone Number |
757-6133
|
| Written Communications |
false
|
| Soliciting Material |
false
|
| Pre-commencement Tender Offer |
false
|
| Pre-commencement Issuer Tender Offer |
false
|
| Entity Emerging Growth Company |
true
|
| Elected Not To Use the Extended Transition Period |
false
|
| Common Class A [Member] |
|
| Document Information [Line Items] |
|
| Title of 12(b) Security |
Class A Ordinary Shares, par value $0.0001 per share
|
| Trading Symbol |
ZURA
|
| Security Exchange Name |
NASDAQ
|
| Warrant [Member] |
|
| Document Information [Line Items] |
|
| Title of 12(b) Security |
Warrants, each whole warrant exercisable for one Class A Ordinary Share at an exercise price of $11.50 per share
|
| Trading Symbol |
ZURAW
|
| Security Exchange Name |
NASDAQ
|
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 2 such as Street or Suite number
| Name: |
dei_EntityAddressAddressLine2 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the state or province.
| Name: |
dei_EntityAddressStateOrProvince |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:stateOrProvinceItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCommission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.
| Name: |
dei_EntityFileNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fileNumberItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 13e
-Subsection 4c
| Name: |
dei_PreCommencementIssuerTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 14d
-Subsection 2b
| Name: |
dei_PreCommencementTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTitle of a 12(b) registered security. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b
| Name: |
dei_Security12bTitle |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:securityTitleItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the Exchange on which a security is registered. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection d1-1
| Name: |
dei_SecurityExchangeName |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarExchangeCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Section 14a
-Number 240
-Subsection 12
| Name: |
dei_SolicitingMaterial |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:tradingSymbolItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Securities Act
-Number 230
-Section 425
| Name: |
dei_WrittenCommunications |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_StatementClassOfStockAxis=us-gaap_CommonClassAMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_StatementClassOfStockAxis=us-gaap_WarrantMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
Grafico Azioni JATT Acquisition (NYSE:JATT)
Storico
Da Mag 2024 a Giu 2024

Grafico Azioni JATT Acquisition (NYSE:JATT)
Storico
Da Giu 2023 a Giu 2024
