HOUSTON, Feb. 23, 2021 /PRNewswire/ -- Soliton, Inc.,
(Nasdaq: SOLY) ("Soliton" or the "Company"), a medical device
company with a novel and proprietary aesthetic platform technology,
today announced that a pre-clinical study in animals demonstrated
positive results for the potential treatment of liver fibrosis.
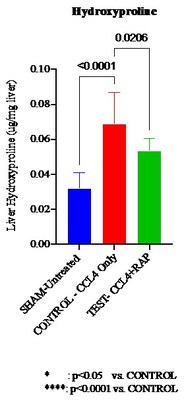
Validated laboratory and histological assessments in a mouse model
demonstrated that Rapid Acoustic Pulse ("RAP") therapy reduced the
effects of induced liver fibrosis 7-days following completion of
carbon tetrachloride (CCL4) induction by 42%. In
addition, post treatment histology slides stained with picrosirius
red (PSR) showed a lower percentage of fibrosis from a single
2-minute RAP treatment compared to the Control group.
"We believe these results represent a significant discovery in
our understanding of RAP's potential," commented Chris Capelli, Vice Chairman and CSO of Soliton.
"Not only does this open up the possibility of addressing a
significant unmet need in the treatment of liver fibrosis, these
results help validate our mechanism of action with both cellulite
and hypertrophic scars, as we believe RAP appears to target the
fibroblast and reduce fibrosis in the tissue."
It is estimated that 6 to 7 percent of the world's population
has liver fibrosis and that many who have it are unaware because
they have no symptoms until it is too late for successful
intervention. Coherent Market Insights Analysis indicates
that the global liver fibrosis treatment market size is expected to
witness a Compounded Annual Growth Rate of 10.8% over the forecast
period (2018 – 2026) reaching a value of US$
28.1 billion in 2026. While researchers are conducting many
tests to try to find medications that can reverse the effects of
liver fibrosis, there are no medications that can accomplish this
currently.
"While we maintain a tight focus on our upcoming commercial
launch of the RAP device in both cellulite reduction and tattoo
removal, we believe exploring new indications with large markets
and significant unmet needs, such as liver fibrosis, offers
potential for increasing long-term shareholder value," said
Brad Hauser, President and CEO of
Soliton. "One of the things that distinguishes Soliton is the
strength of the science that underlies RAP. This latest research
underscores just how unique our acoustic shockwave technology is
and how potentially important its applications could be. We
look forward to continuing to examine the strong promise the RAP
technology offers across many fibrotic conditions."
The RAP device is currently cleared by the FDA for temporary
improvement in the appearance of cellulite. The RAP device is also
indicated for use as an accessory to the 1064 nm Q-Switched laser
for black ink tattoo removal in Fitzpatrick Skin Type I-III
patients. The technology is not cleared for the treatment of liver
fibrosis. The Company plans to begin selling the device for both
tattoo removal and cellulite treatment in the first half of
2021.
Join our more than 200K
subscribers here to follow the
Company: https://soly-investors.com
About Soliton, Inc.
Soliton, Inc. is a medical device company with a novel and
proprietary platform technology licensed from The University of Texas on behalf of MD Anderson Cancer
Center. The Company's first FDA cleared commercial product will use
rapid pulses of acoustic shockwaves as an accessory to lasers for
the removal of unwanted tattoos and the treatment of cellulite. The
Company is based in Houston,
Texas, and is actively engaged in bringing the Rapid
Acoustic Pulse ("RAP") device to the market. The Company believes
this "Soliton" method has the potential to lower tattoo removal
costs for patients, while increasing profitability to
practitioners, compared to current laser removal methods. The
Company also believe the technology will provide the first
non-invasive acoustic technology to target the underlying causes of
dimples and ridges in cellulite. Soliton is investigating potential
additional capabilities of the RAP technology. The device is
currently cleared in the United
States only for use in tattoo removal and cellulite.
For more information about the Company, please
visit: http://www.soliton.com
Forward-Looking Statements
This press release includes forward-looking statements within
the meaning of Section 27A of the Securities Act of 1933, Section
21E of the Securities Exchange Act of 1934 and the Private
Securities Litigation Reform Act of 1995, which statements involve
risks and uncertainties. Forward-looking statements in this press
release include, without limitation, our ability to successfully
replicate the results of this animal study in further clinical
studies in either animals or humans and to launch our technology in
the months to come. These statements relate to future events,
future expectations, plans and prospects. Although Soliton believes
that the expectations reflected in such forward-looking statements
are reasonable as of the date made, actual results or outcomes may
prove to be materially different from the expectations expressed or
implied by such forward-looking statements. Soliton has attempted
to identify forward-looking statements by terminology including
''believes,'' ''estimates,'' ''anticipates,'' ''expects,''
''plans,'' ''projects,'' ''intends,'' ''potential,'' ''may,''
''could,'' ''might,'' ''will,'' "would," ''should,''
''approximately'' or other words that convey uncertainty of future
events or outcomes to identify these forward-looking statements.
These statements are only predictions and involve known and unknown
risks, uncertainties, and other factors, including those discussed
in our filings with the Securities and Exchange Commission ("SEC"),
including under the heading " Risk Factors" in our most recently
filed Form 10-K filed with the SEC and as updated in our Form 10-Q
filings and in our other filings with the SEC. Any
forward-looking statements contained in this release speak only as
of its date. Soliton undertakes no obligation to update any
forward-looking statements contained in this release to reflect
events or circumstances occurring after its date or to reflect the
occurrence of unanticipated events.

 View original content to download
multimedia:http://www.prnewswire.com/news-releases/soliton-announces-rap-treatment-reduces-effects-of-liver-fibrosis-by-42-in-animal-study-301232950.html
View original content to download
multimedia:http://www.prnewswire.com/news-releases/soliton-announces-rap-treatment-reduces-effects-of-liver-fibrosis-by-42-in-animal-study-301232950.html
SOURCE Soliton, Inc.