CARMAT : Availability of the 2023 Universal Registration Document
30 Aprile 2024 - 6:00PM
Business Wire
Regulatory News:
CARMAT (FR0010907956, ALCAR), designer and developer of the
world’s most advanced total artificial heart, aiming to provide a
therapeutic alternative for people suffering from advanced
biventricular heart failure (the “Company” or
“CARMAT”), today announced the publication of the Company’s
Universal registration document (“URD”) for the year ended December
31, 2023.
The document, filed with the French stock-market authority
(Autorité des Marchés Financiers) on April 30, 2023, is available
to the public free of charge upon request, as per current legal
regulations; and on the Company’s website under the section
Investors / Documentation, as well as on that of the AMF
(www.amf-france.org).
It notably includes the 2023 annual financial report, the report
on corporate governance, the required information in relation to
the share repurchase program, as well as the statutory auditor’
reports and information on the fees paid to the statutory auditor
in 2023. The URD is available in French only, but for the
convenience of English-speaking readers, CARMAT provides a free
translation into English of its 2023 financial statements1, which
is available in the English section of its website.
●●●
About CARMAT
CARMAT is a French MedTech that designs, manufactures and
markets the Aeson® artificial heart. The Company’s ambition is to
make Aeson® the first alternative to a heart transplant, and thus
provide a therapeutic solution to people suffering from end-stage
biventricular heart failure, who are facing a well-known shortfall
in available human grafts. The world’s first physiological
artificial heart that is highly hemocompatible, pulsatile and
self-regulated, Aeson® could save, every year, the lives of
thousands of patients waiting for a heart transplant. The device
offers patients quality of life and mobility thanks to its
ergonomic and portable external power supply system that is
continuously connected to the implanted prosthesis. Aeson® is
commercially available as a bridge to transplant in the European
Union and other countries that recognize CE marking. Aeson® is also
currently being assessed within the framework of an Early
Feasibility Study (EFS) in the United States. Founded in 2008,
CARMAT is based in the Paris region, with its head offices located
in Vélizy-Villacoublay and its production site in Bois-d’Arcy. The
Company can rely on the talent and expertise of a multidisciplinary
team of circa 200 highly specialized people. CARMAT is listed on
the Euronext Growth market in Paris (Ticker: ALCAR / ISIN code:
FR0010907956).
For more information, please go to www.carmatsa.com and follow
us on LinkedIn.
Name: CARMAT ISIN code:
FR0010907956 Ticker: ALCAR
Disclaimer
This press release and the information contained herein do not
constitute an offer to sell or subscribe, nor a solicitation of an
order to buy or subscribe to CARMAT shares in any country. This
press release may contain forward-looking statements by the company
regarding its objectives and prospects. These forward-looking
statements are based on the current estimates and anticipations of
the company's management and are subject to risk factors and
uncertainties such as the company's ability to implement its
strategy, the pace of development of CARMAT's production and sales,
the pace and results of ongoing or planned clinical trials,
technological evolution and competitive environment, regulatory
changes, industrial risks, and all risks associated with the
company's growth management. The company's objectives mentioned in
this press release may not be achieved due to these elements or
other risk factors and uncertainties.
Significant and specific risks of the company are those
described in its universal registration document filed with the
French Financial Markets Authority (Autorité des marchés financiers
- the “AMF”) under number D.23-0374. Readers' attention is
particularly drawn to the fact that the company's current cash
runway is limited to mid-May 2024. Readers and investors are also
advised that other risks, unknown or not considered significant and
specific, may or could exist.
Aeson® is an active implantable medical device commercially
available in the European Union and other countries recognizing CE
marking. The Aeson® total artificial heart is intended to replace
the ventricles of the native heart and is indicated as a bridge to
transplant for patients suffering from end-stage biventricular
heart failure (INTERMACS classes 1-4) who cannot benefit from
maximal medical therapy or a left ventricular assist device (LVAD)
and who are likely to undergo a heart transplant within 180 days of
implantation. The decision to implant and the surgical procedure
must be carried out by healthcare professionals trained by the
manufacturer. The documentation (clinician manual, patient manual,
and alarm booklet) should be carefully read to understand the
features of Aeson® and the information necessary for patient
selection and proper use (contraindications, precautions, side
effects). In the United States, Aeson® is currently exclusively
available as part of an Early Feasibility Study approved by the
Food & Drug Administration (FDA).
1 Corresponding to Section 3.2 of the URD.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20240430237833/en/
CARMAT Stéphane Piat Chief Executive Officer
Pascale d’Arbonneau Chief Financial Officer Tel.: +33 1
39 45 64 50 contact@carmatsas.com
Alize RP Press Relations
Caroline Carmagnol Tel.: +33 6 64 18 99 59
carmat@alizerp.com
NewCap Financial Communication & Investor
Relations
Dusan Oresansky Jérémy Digel Tel.: +33 1 44 71 94
92 carmat@newcap.eu
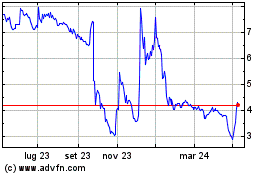
Grafico Azioni Carmat (EU:ALCAR)
Storico
Da Apr 2024 a Mag 2024
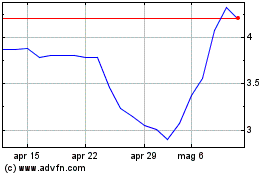
Grafico Azioni Carmat (EU:ALCAR)
Storico
Da Mag 2023 a Mag 2024