false
0001850119
0001850119
2023-12-09
2023-12-09
iso4217:USD
xbrli:shares
iso4217:USD
xbrli:shares
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, DC 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported):
December 9, 2023
Century Therapeutics, Inc.
(Exact name of registrant as specified in its charter)
| Delaware |
|
001-40498 |
|
84-2040295 |
(State or other jurisdiction of
incorporation or organization) |
|
(Commission File Number) |
|
(I.R.S. Employer
Identification No.) |
|
25
North 38th Street, 11th Floor
Philadelphia, Pennsylvania |
|
19104 |
| (Address of principal executive offices) |
|
(Zip Code) |
Registrant’s telephone number, including
area code: (267) 817-5790
Not Applicable
(Former name or former address, if changed since
last report)
Check the appropriate box below if the Form 8-K filing is intended
to simultaneously satisfy the filing obligation of the registrant under any of the following provisions (see General Instruction A.2.
below):
| ¨ |
Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| ¨ |
Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| ¨ |
Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| ¨ |
Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
| Title of Each Class |
|
Trading Symbol |
|
Name
of Exchange on Which Registered |
| Common Stock, par value $0.0001 per share |
|
IPSC |
|
Nasdaq Global Select Market |
Indicate by check mark whether the registrant is an emerging growth
company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange
Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company x
If an emerging growth company, indicate by check mark if the registrant
has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant
to Section 13(a) of the Exchange Act.
On December 9, 2023, Century Therapeutics, Inc.
(the “Company”) issued a press release announcing the presentation of initial clinical data from a single-patient case study
in which the Company believes support the potential for a multi-dosing strategy for CAR iNK enabled by Allo-Evasion™ edits at the
65th American Society of Hematology Annual Meeting and Exposition, being held December 9-12th in San Diego,
California. A copy of the press release is furnished as Exhibit 99.1 to this Current Report on Form 8-K and is incorporated
herein by reference.
On December 11, 2023, the Company updated
information reflected in a slide presentation, which is attached as Exhibit 99.2 to this Current Report on Form 8-K and is incorporated
herein by reference. Representatives of the Company will use the updated presentation in various meetings with investors from time to
time.
| Item 9.01 |
Financial Statements and Exhibits |
(d) Exhibits
SIGNATURES
Pursuant to the requirements of the Securities
Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| |
CENTURY THERAPEUTICS, INC. |
| |
|
|
| |
By: |
/s/ Brent Pfeiffenberger, Pharm.D. |
| |
Name: |
Brent Pfeiffenberger, Pharm.D. |
| |
Title: |
President and Chief Executive Officer |
Date: December 11, 2023
Exhibit 99.1

Century
Therapeutics Presents Initial Data from CNTY-101 Phase 1 ELiPSE-1 Trial Supporting the Potential
for a Multi-Dosing Strategy for CAR iNK Enabled by Allo-Evasion™ Edits
– Data presented at 65th ASH
Annual Meeting show CNTY-101 was generally well tolerated at Dose Level 1 (100 million cells) in a high-risk, heavily pretreated R/R B-cell
lymphoma patient –
–
Preliminary clinical data demonstrate six-month durable complete response in Dose Level 1 in a single patient following
multiple cycles of CNTY-101 without lymphodepletion –
–
Pharmacokinetic data suggests CNTY-101 exposure may be maintained upon administration of additional cycles without lymphodepletion
due to lack of observed allo-rejection –
–
Company to host conference call on Monday, December 11 at 7:30 AM PT/10:30 AM ET to review ASH data including additional
clinical results from Dose Level 1 (100 million cells) and Dose Level 2 (300 million cells), as well as clinical plans for CNTY-101 in
systemic lupus erythematosus –
PHILADELPHIA,
December 9, 2023 – Century Therapeutics (NASDAQ: IPSC), an innovative biotechnology company developing
induced pluripotent stem cell (iPSC)-derived cell therapies in immuno-oncology and autoimmune and inflammatory disease, today announced
the presentation of initial clinical data from a single-patient case study which Century believes support the potential for a multi-dosing
strategy for CAR iNK enabled by Allo-Evasion™ edits at the 65th American Society of Hematology (ASH) Annual Meeting and Exposition,
being held December 9-12 in San Diego. The poster, titled, “Multiple Doses of CNTY-101, an iPSC-Derived Allogeneic CD19 Targeting
CAR-NK Product, are Safe and Result in Tumor Microenvironment Changes Associated with Response: A Case Study”, is available on
the Scientific Resources page of the Company’s website.
“We are thrilled that the initial clinical
evidence for CNTY-101 provides support for the potential for Allo-Evasion™ to enable a multi-dosing regimen without the need for
continued lymphodepletion. This is highly encouraging in advancing our goal to increase persistence of the cells during the treatment
period and potentially lead to deeper and more durable responses,” said Brent Pfeiffenberger, Pharm.D., Chief Executive Officer
of Century Therapeutics. “We look forward to advancing the study at both higher and more frequent doses of CNTY-101, and plan to
present additional clinical data in mid-2024.”
“As
the first cell therapy product candidate engineered with six precision gene edits aimed at providing selectivity and persistence, CNTY-101
is positioned to potentially fill a high unmet need among heavily pretreated non-Hodgkin lymphoma patients,” said Krish Patel, M.D.,
Director of Lymphoma Program, Director of Hematologic Malignancies and Cellular Therapy, Swedish Cancer Institute, Seattle. “The
encouraging initial data presented today from this patient who received low
doses of CNTY-101 exhibits signals of persistence of CNTY-101 cells out of circulation and supports testing at higher doses. I look forward
to the continuation of the study and to further investigating the full therapeutic potential of CNTY-101.”

Data featured in a single-patient case study presented
at ASH involves a 63-year-old patient with relapsed/refractory (R/R) progressive follicular lymphoma previously treated with four prior
lines of therapy who was enrolled at Dose Level 1 (100 million cells). As of a data cutoff date of November 13, 2023, the patient
has received seven 28-day cycles of a single infusion of CNTY-101 at Dose Level 1. Cycles one and two included three days of lymphodepletion
(LD), whereas cycles three through seven were given with no LD. Interleukin-2 (IL-2) was administered for all cycles except for the first.
The patient maintained a complete response with a duration of six months before subsequently progressing.
Data from the single-patient case study indicated
that CNTY-101 was generally well tolerated in this patient at Dose level 1 (100 million cells). No dose-limiting toxicities, cytokine
release syndrome or immune effector cell–associated neurotoxicity syndrome were observed, and no adverse events related to treatment
with CNTY-101 were detected in this patient, to date. Additionally, no concerted changes in inflammatory cytokines and mediators associated
with cytokine release syndrome or neurotoxicity have been detected in this patient.
Following administration of two cycles with and
three cycles without LD, serum assessments from available data of the first five cycles of CNTY-101 treatment in this patient showed no
evidence of functional pre-existing or induced humoral immunogenicity against CNTY-101. Importantly, tumor microenvironment initial analyses
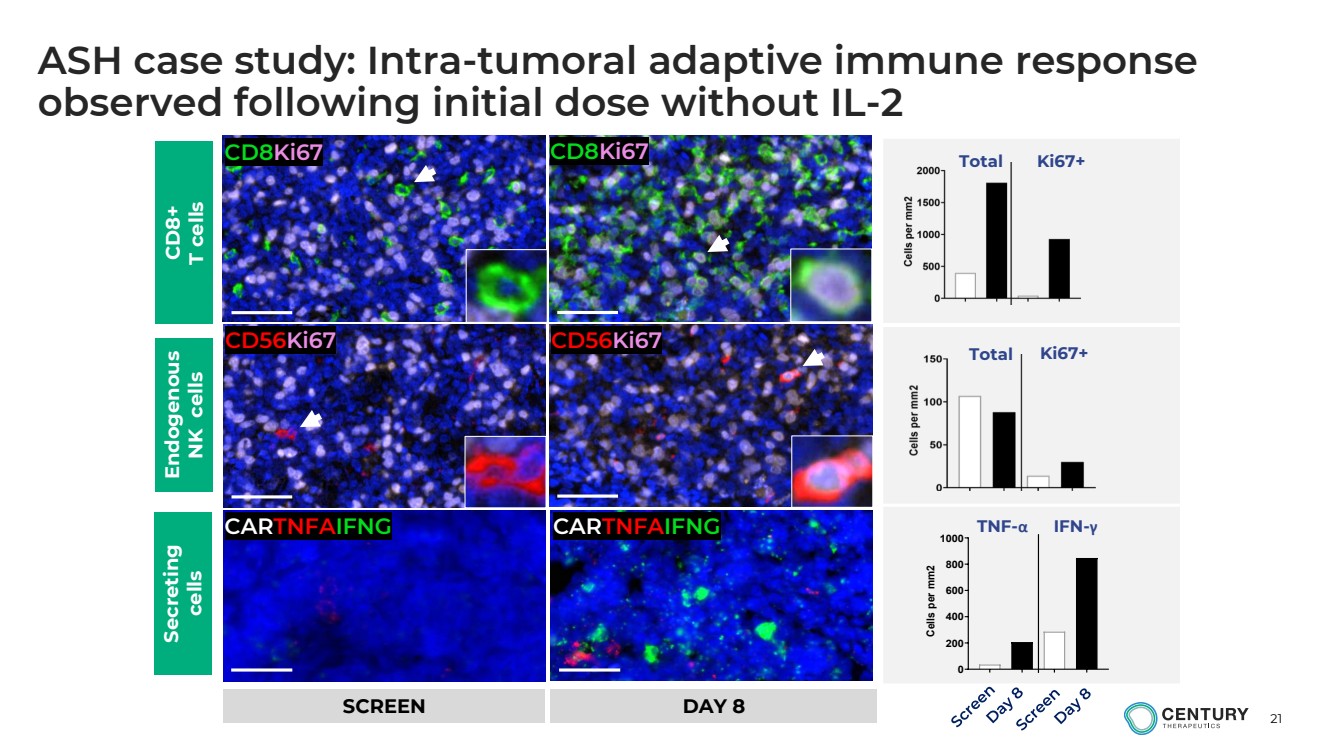
demonstrated a vigorous increase in T cells within 8 days of the 1st CNTY-101 cell infusion. Increases in proliferating cytotoxic
T cells and TNFα and IFNγ-secreting cells were observed, suggestive of induction of adaptive immune responses within the tumor.
Additionally, ddPCR analysis of CNTY-101 genomic DNA and cell-free DNA from Dose Level 1 patient (n=4) samples suggest that CNTY-101 cells
were able to traffic out of circulation shortly after infusion and showed persistence in tissues for at least 3 days.
In addition to the preliminary clinical data presented
today, the Company will also present additional results from patients treated at Dose Level 1 (100 million cell dose), as well as preliminary
data from three patients treated at Dose Level 2 (300 million cell dose) during a conference call and webcast on Monday, December 11
at 7:30 AM PT/10:30 AM ET. In addition, the Company will discuss its planned Phase 1 trial, including supporting preclinical data, for
CNTY-101 in systemic lupus erythematosus, the Company’s first autoimmune and inflammatory disease indication.
Conference Call and Webcast
The
live audio webcast and accompanying slides may be accessed through the Events & Presentations page in
the Investors section of the Company's website. Alternatively, the conference call may be accessed through the following:
| · | Conference ID: century2023 |
| · | Domestic Dial-in Number: (800) 590-8290 |
| · | International Dial-in Number: (240) 690-8800 |
| · | Live
webcast: https://century-therapeutics-initial-clinical-data-call.open-exchange.net/ |
For
those unable to participate in the conference call or webcast, a replay will be available on the Investors section of the Company's website
at www.centurytx.com approximately 24 hours after the conference call and will be available for 90 days following
the call.

About Allo-Evasion™
Century’s proprietary Allo-Evasion™
technology is used to engineer cell therapy product candidates with the potential to evade identification by the host immune system so
they can be dosed multiple times without rejection, enabling increased persistence of the cells during the treatment period and potentially
leading to deeper and more durable responses. More specifically, Allo-Evasion™ 1.0 technology incorporates three gene edits designed
to avoid recognition by patient/host CD8+ T cells, CD4+ T cells and NK cells. Knockout of beta-2-microglobulin or β2m, designed to
prevent CD8+ T cell recognition, knock-out of the Class II Major Histocompatibility Complex Transactivator, or CIITA, designed to
prevent CD4+ T cell recognition, and knock-in of the HLA-E gene, designed to enable higher expression of the HLA-E protein to prevent
killing of CNTY-101 cells by host NK cells. Allo-Evasion™ technology may allow the implementation of more flexible and effective
repeat dosing protocols for off-the-shelf product candidates.
About ELiPSE-1
The
Phase 1 ELiPSE-1 trial (NCT05336409) is intended to assess the safety, tolerability, pharmacokinetics, and preliminary
efficacy of CNTY-101 in adult patients with relapsed or refractory CD19-positive B-cell lymphomas. All patients will receive an initial
standard dose of conditioning chemotherapy consisting of cyclophosphamide (300 mg/m2) and fludarabine (30mg/m2) for 3 days. Schedule
A of the trial includes a single-dose escalation of CNTY-101 and subcutaneous IL-2. Schedule B will evaluate a three-dose schedule per
cycle of CNTY-101. Patients who demonstrate a clinical benefit are eligible for additional cycles of treatment with or without additional
lymphodepletion.
About CNTY-101
CNTY-101 is an investigational off-the-shelf immunotherapy
product candidate that utilizes iPSC-derived natural killer (NK) cells with a CD19-directed chimeric antigen receptor (CAR) and includes
Century’s core Allo-Evasion™ edits designed to overcome the three major pathways of host versus graft rejection - CD8+ T cells,
CD4+ T cells and NK cells. In addition, the product candidate is engineered to express IL-15 to provide homeostatic cytokine support,
which has been shown pre-clinically to improve functionality and persistence. Further, to potentially improve safety, the iNK cells were
engineered with an EGFR safety switch, and proof-of-concept studies have demonstrated that the cells can be quickly eliminated by the
administration of cetuximab, an antibody against EGFR approved by the U.S. Food and Drug Administration for certain cancers. Century is
currently assessing CNTY-101 in patients with relapsed or refractory CD19-positive B-cell lymphomas in its Phase 1 ELiPSE-1 clinical trial
and intends to initiate its second Phase 1 clinical trial assessing CNTY-101 in patients with moderate to severe systemic lupus erythematosus.
About Century Therapeutics
Century
Therapeutics (NASDAQ: IPSC) is harnessing the power of adult stem cells to develop curative cell therapy products for cancer and autoimmune
and inflammatory diseases that we believe will allow us to overcome the limitations of first-generation cell therapies. Our genetically
engineered, iPSC-derived cell product candidates are designed to specifically target hematologic and solid tumor cancers, with a broadening
application to autoimmune and inflammatory diseases. We are leveraging our expertise in cellular reprogramming, genetic engineering,
and manufacturing to develop therapies with the potential to overcome many of the challenges inherent to cell therapy and provide a significant
advantage over existing cell therapy technologies. We believe our commitment to developing off-the-shelf cell therapies will expand patient
access and provide an unparalleled opportunity to advance the course of cancer and autoimmune and inflammatory disease care. For more
information on Century Therapeutics please visit www.centurytx.com.

Century Therapeutics Forward-Looking Statement
This press release contains forward-looking statements
within the meaning of, and made pursuant to the safe harbor provisions of, The Private Securities Litigation Reform Act of 1995. All statements
contained in this press release, other than statements of historical facts or statements that relate to present facts or current conditions,
including but not limited to, statements regarding our clinical development plans and timelines, are forward-looking statements. These
statements involve known and unknown risks, uncertainties and other important factors that may cause our actual results, performance,
or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking
statements. In some cases, you can identify forward-looking statements by terms such as “may,” “might,” “will,”
“should,” “expect,” “plan,” “aim,” “seek,” “anticipate,” “could,”
“intend,” “target,” “project,” “contemplate,” “believe,” “estimate,”
“predict,” “forecast,” “potential” or “continue” or the negative of these terms or other
similar expressions. The forward-looking statements in this press release are only predictions. We have based these forward-looking statements
largely on our current expectations and projections about future events and financial trends that we believe may affect our business,
financial condition, and results of operations. These forward-looking statements speak only as of the date of this press release and are
subject to a number of risks, uncertainties and assumptions, some of which cannot be predicted or quantified and some of which are beyond
our control, including, among others: our ability to successfully advance our current and future product candidates through development
activities, preclinical studies, and clinical trials; our dependence on the success of our lead product candidate, CNTY-101; the ability
of CNTY-101 to be administered as part of a multi-dose strategy and to enable responses without lymphodepletion; uncertainties inherent
in the results of preliminary data, pre-clinical studies and earlier-stage clinical trials, which may not be predictive of final results
or the results of later-stage clinical trials; the timing of and our ability to initiate and successfully enroll the Phase 1 SLE trial;
our ability to obtain FDA clearance of our future IND submissions and commence and complete clinical trials on expected timelines, or
at all; our reliance on the maintenance of certain key collaborative relationships for the manufacturing and development of our product
candidates; the timing, scope and likelihood of regulatory filings and approvals, including final regulatory approval of our product candidates;
the impact of geopolitical issues, banking instability and inflation on our business and operations, supply chain and labor force; the
performance of third parties in connection with the development of our product candidates, including third parties conducting our clinical
trials as well as third-party suppliers and manufacturers; our ability to successfully commercialize our product candidates and develop
sales and marketing capabilities, if our product candidates are approved; our ability to recruit and maintain key members of management
and our ability to maintain and successfully enforce adequate intellectual property protection. These and other risks and uncertainties
are described more fully in the “Risk Factors” section of our most recent filings with the Securities and Exchange Commission
and available at www.sec.gov. You should not rely on these forward-looking statements as predictions of future events. The events and
circumstances reflected in our forward-looking statements may not be achieved or occur, and actual results could differ materially from
those projected in the forward-looking statements. Moreover, we operate in a dynamic industry and economy. New risk factors and uncertainties
may emerge from time to time, and it is not possible for management to predict all risk factors and uncertainties that we may face. Except
as required by applicable law, we do not plan to publicly update or revise any forward-looking statements contained herein, whether as
a result of any new information, future events, changed circumstances or otherwise.

For More Information:
Investors/Media: Melissa Forst/Maghan Meyers
– century@argotpartners.com
Exhibit 99.2
| Initial Data from Phase 1 ELiPSE-1 Trial
of CNTY-101 in Relapsed/Refractory B-cell Lymphomas and Overview of
Planned Phase 1 Study in Systemic
Lupus Erythematosus
December 11, 2023 |

| 2
Forward-looking statements
This presentation contains forward-looking statements within the meaning of, and made pursuant to the safe harbour provisions of, The Private Securities
Litigation Reform Act of 1995. All statements contained in this document, other than statements of historical facts or statements that relate to present facts or
current conditions, including but not limited to, statements regarding possible or assumed future results of operations, business strategies, research and
development plans, regulatory activities, market opportunity, competitive position and potential growth opportunities are forward-looking statements. These
statements involve known and unknown risks, uncertainties and other important factors that may cause our actual results, performance or achievements to be
materially different from any future results, performance or achievements expressed or implied by the forward-looking statements. In some cases, you can
identify forward-looking statements by terms such as “may,” “might,” “will,” “should,” “expect,” “plan,” “aim,” “seek,” “anticipate,” “could,” “intend,” “target,”
“project,” “contemplate,” “believe,” “estimate,” “predict,” “forecast,” “potential” or “continue” or the negative of these terms or other similar expressions. The
forward-looking statements in this presentation are only predictions. We have based these forward-looking statements largely on our current expectations and
projections about future events and financial trends that we believe may affect our business, financial condition and results of operations. These forward-looking
statements speak only as of the date of this presentation and are subject to a number of risks, uncertainties and assumptions, some of which cannot be
predicted or quantified and some of which are beyond our control, including, among others: our ability to successfully advance our current and future product
candidates through development activities, preclinical studies, and clinical trials; our dependence on the success of our lead product candidate, CNTY-101; the
ability of CNTY-101 to be administered as part of a multi-dose strategy and to enable responses without lymphodepletion; uncertainties inherent in the results of
preliminary data, pre-clinical studies and earlier-stage clinical trials, which may not be predictive of final results or the results of later-stage clinical trials; the
timing of and our ability to initiate and successfully enroll the Phase 1 SLE trial; our ability to obtain FDA clearance of our future IND submissions and commence
and complete clinical trials on expected timelines, or at all; our reliance on the maintenance on certain key collaborative relationships for the manufacturing
and development of our product candidates; the timing, scope and likelihood of regulatory filings and approvals, including final regulatory approval of our
product candidates; the impact of, geopolitical issues and inflation on our business and operations, supply chain and labor force; the performance of third
parties in connection with the development of our product candidates, including third parties conducting our future clinical trials as well as third-party suppliers
and manufacturers; our ability to successfully commercialize our product candidates and develop sales and marketing capabilities, if our product candidates are
approved; and our ability to maintain and successfully enforce adequate intellectual property protection. These and other risks and uncertainties are described
more fully in the “Risk Factors” section of our most recent filings with the Securities and Exchange Commission and available at www.sec.gov. You should not
rely on these forward-looking statements as predictions of future events. The events and circumstances reflected in our forward-looking statements may not be
achieved or occur, and actual results could differ materially from those projected in the forward-looking statements. Moreover, we operate in a dynamic industry
and economy. New risk factors and uncertainties may emerge from time to time, and it is not possible for management to predict all risk factors and
uncertainties that we may face. Except as required by applicable law, we do not plan to publicly update or revise any forward-looking statements contained
herein, whether as a result of any new information, future events, changed circumstances or otherwise. |

| 3
Today’s agenda
• Introduction
Brent Pfeiffenberger, Pharm.D., Chief Executive Officer
• Overview of Foundational Platform Technologies
Hy Levitsky, M.D., President of Research and Development
• Review of Initial ELiPSE-1 Data for CNTY-101
Nick Trede, M.D., Ph.D., SVP, Head of Clinical Development
• CNTY-101 in Systemic Lupus Erythematosus
Adrienne Farid, Ph.D., Chief Operations Officer and Head of Early Development
• Closing
Brent Pfeiffenberger, Pharm.D., Chief Executive Officer
• Also Joining for Q&A
Michael Diem, M.D., Chief Financial Officer
Greg Russotti, Ph.D., Chief Technology and Manufacturing Officer |

| 4
Century Therapeutics: Building an industry-leading, next-generation allogeneic iPSC-derived cell therapy platform
LIMITLESS POTENTIAL...
PRECISION DESIGN...
ENDURING IMPACT...
Foundational investments in iPSC technology,
genetic editing, and manufacturing
Progressing multiple clinical programs in
oncology and autoimmune and inflammatory
diseases
Well-capitalized into 2026 to enable delivery
on key milestones and clinical data |

| Overview of Foundational
Platform Technologies:
iPSCs, Allo-EvasionTM and
the Creation of CNTY-101 |
| 6
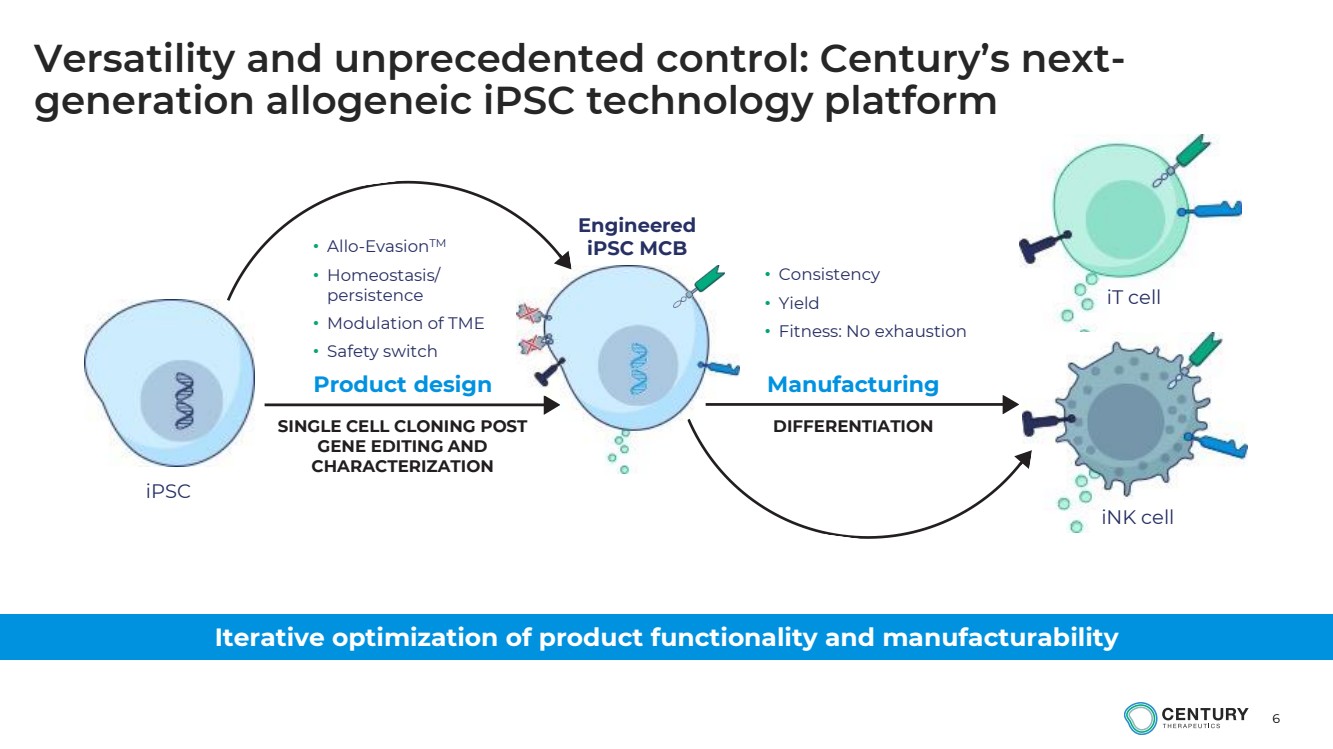
Versatility and unprecedented control: Century’s next-generation allogeneic iPSC technology platform
Iterative optimization of product functionality and manufacturability
• Consistency
• Yield
• Fitness: No exhaustion
Product design Manufacturing
iPSC
iT cell
iNK cell
Engineered
iPSC MCB
SINGLE CELL CLONING POST
GENE EDITING AND
CHARACTERIZATION
DIFFERENTIATION
• Allo-EvasionTM
• Homeostasis/
persistence
• Modulation of TME
• Safety switch |
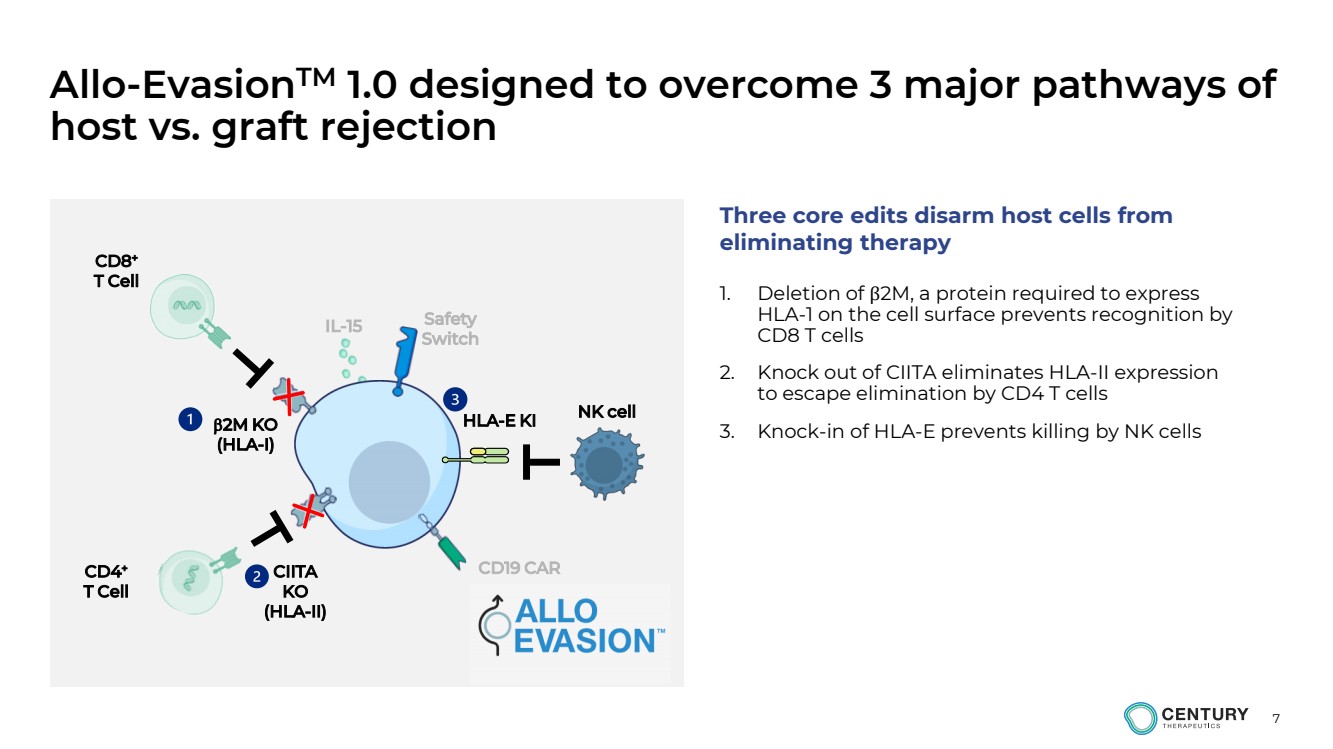
| 7
Allo-EvasionTM 1.0 designed to overcome 3 major pathways of
host vs. graft rejection
1. Deletion of β2M, a protein required to express
HLA-1 on the cell surface prevents recognition by
CD8 T cells
2. Knock out of CIITA eliminates HLA-II expression
to escape elimination by CD4 T cells
3. Knock-in of HLA-E prevents killing by NK cells 2M KO
(HLA-I)
HLA-E KI
CIITA
KO
(HLA-II)
CD8+
T Cell
CD4+
T Cell
NK cell
Three core edits disarm host cells from
eliminating therapy
CD19 CAR
Safety
Switch IL-15 |
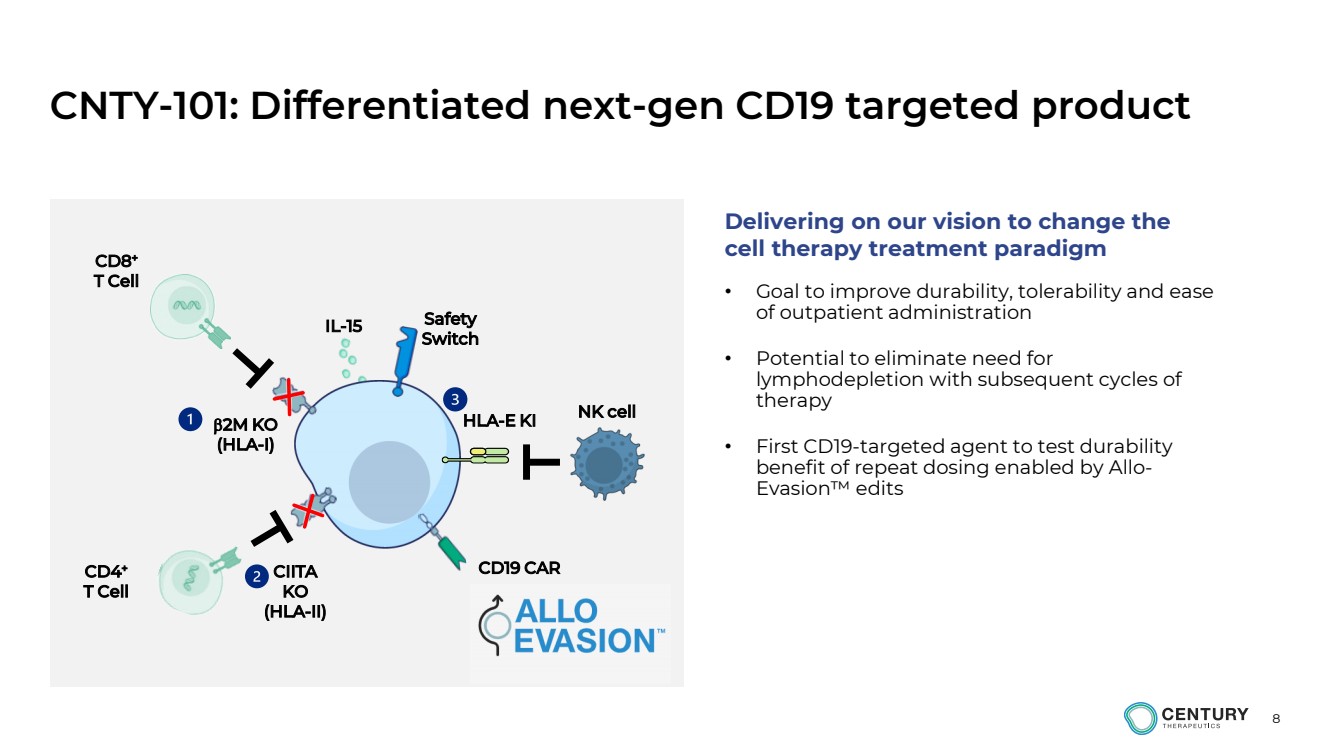
| 8
CNTY-101: Differentiated next-gen CD19 targeted product
2M KO
(HLA-I)
HLA-E KI
CIITA
KO
(HLA-II)
CD8+
T Cell
CD4+
T Cell
NK cell
CD19 CAR
Safety
Switch IL-15
• Goal to improve durability, tolerability and ease
of outpatient administration
• Potential to eliminate need for
lymphodepletion with subsequent cycles of
therapy
• First CD19-targeted agent to test durability
benefit of repeat dosing enabled by Allo-Evasion™ edits
Delivering on our vision to change the
cell therapy treatment paradigm |
| 9
CNTY-101: Extending drug exposure in R/R B-cell NHL via
repeat dosing and changing the treatment paradigm
with Allo-EvasionTM
R/R: relapsed or refractory, NHL: non-Hodgkin lymphoma, CAR-T: chimeric antigen receptor T cell therapy
Potential solution from Century’s platform:
• Off-the-shelf product offers immediate access
and consistency
• Multiple doses to increase pharmacological pressure to
increase durability
• Host rejection addressed by Allo-EvasionTM edits
Unmet need:
• Autologous CD19 CAR-T curative in only a subset
of patients
• Autologous CD19 CAR-T access is limited and/or can fail in
manufacturing as quality is dependent on patient-derived starting material
• Limited options and poor prognosis for patients who fail
autologous CAR-T
Aim: extending the period of pharmacologic pressure on tumor cells |

| Review of Initial ELiPSE-1 Data
for CNTY-101 |
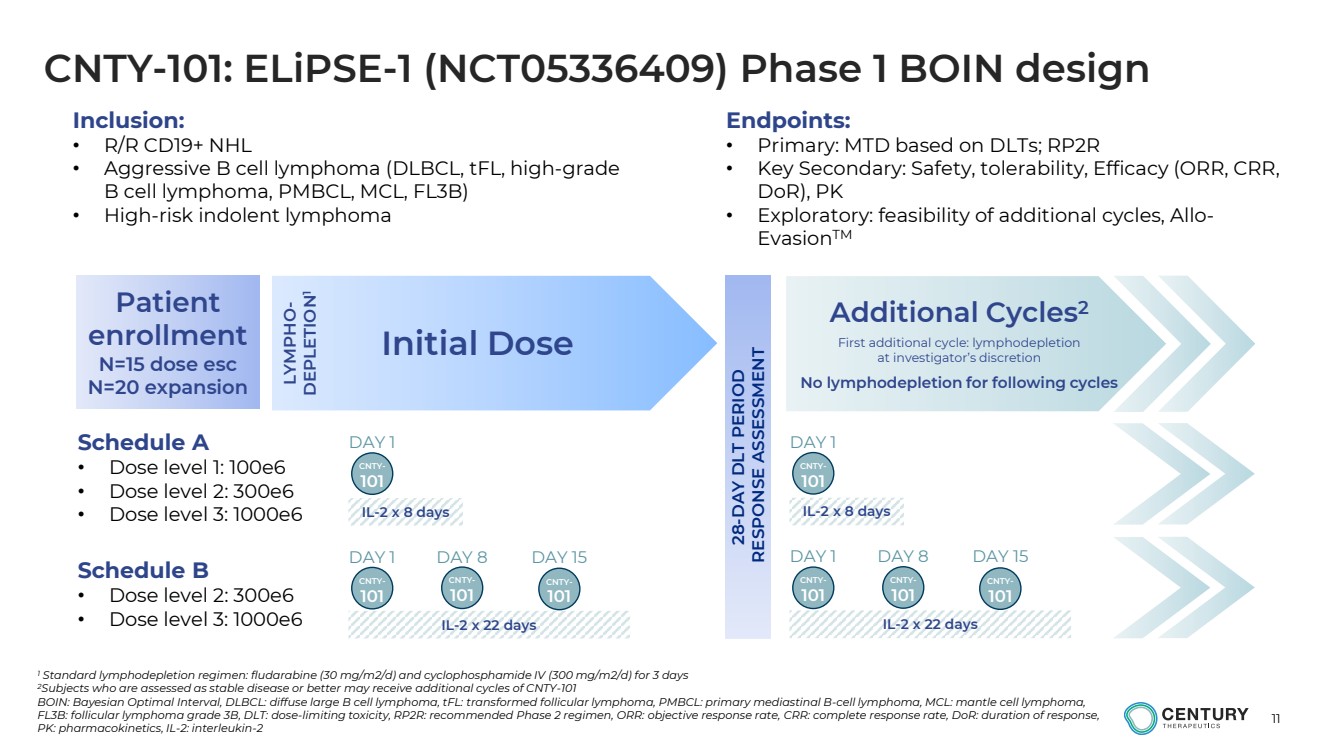
| 11
CNTY-101: ELiPSE-1 (NCT05336409) Phase 1 BOIN design
1 Standard lymphodepletion regimen: fludarabine (30 mg/m2/d) and cyclophosphamide IV (300 mg/m2/d) for 3 days
2Subjects who are assessed as stable disease or better may receive additional cycles of CNTY-101
BOIN: Bayesian Optimal Interval, DLBCL: diffuse large B cell lymphoma, tFL: transformed follicular lymphoma, PMBCL: primary mediastinal B-cell lymphoma, MCL: mantle cell lymphoma,
FL3B: follicular lymphoma grade 3B, DLT: dose-limiting toxicity, RP2R: recommended Phase 2 regimen, ORR: objective response rate, CRR: complete response rate, DoR: duration of response,
PK: pharmacokinetics, IL-2: interleukin-2
Initial Dose
Additional Cycles2
First additional cycle: lymphodepletion
at investigator’s discretion
No lymphodepletion for following cycles
Schedule A
• Dose level 1: 100e6
• Dose level 2: 300e6
• Dose level 3: 1000e6LYMPHO-DEPLETION1
IL-2 x 22 days
Schedule B
• Dose level 2: 300e6
• Dose level 3: 1000e6
28-DAY DLT PERIOD
RESPONSE ASSESSMENT
Patient
enrollment
N=15 dose esc
N=20 expansion
CNTY-101
DAY 1
DAY 1 DAY 8 DAY 15
IL-2 x 8 days
CNTY-101
CNTY-101
CNTY-101
IL-2 x 22 days
CNTY-101
DAY 1
DAY 1 DAY 8 DAY 15
IL-2 x 8 days
CNTY-101
CNTY-101
CNTY-101
Inclusion:
• R/R CD19+ NHL
• Aggressive B cell lymphoma (DLBCL, tFL, high-grade
B cell lymphoma, PMBCL, MCL, FL3B)
• High-risk indolent lymphoma
Endpoints:
• Primary: MTD based on DLTs; RP2R
• Key Secondary: Safety, tolerability, Efficacy (ORR, CRR,
DoR), PK
• Exploratory: feasibility of additional cycles, Allo-EvasionTM |

| 12
ELiPSE-1 initial data: Key takeaways
• Heavily pretreated and refractory patient population treated in first-in-human dose escalation trial
• Favorable safety profile; can be delivered in an outpatient setting
• Encouraging early efficacy signals at lowest dose levels
o 2 patients achieving CR, including 1 patient with 6-month durable CR
• No evidence of allo-rejection
• Initial data for CNTY-101 supports the potential for Allo-Evasion™ to enable a multi-dosing regimen without the need for continued lymphodepletion
CR: Complete response |
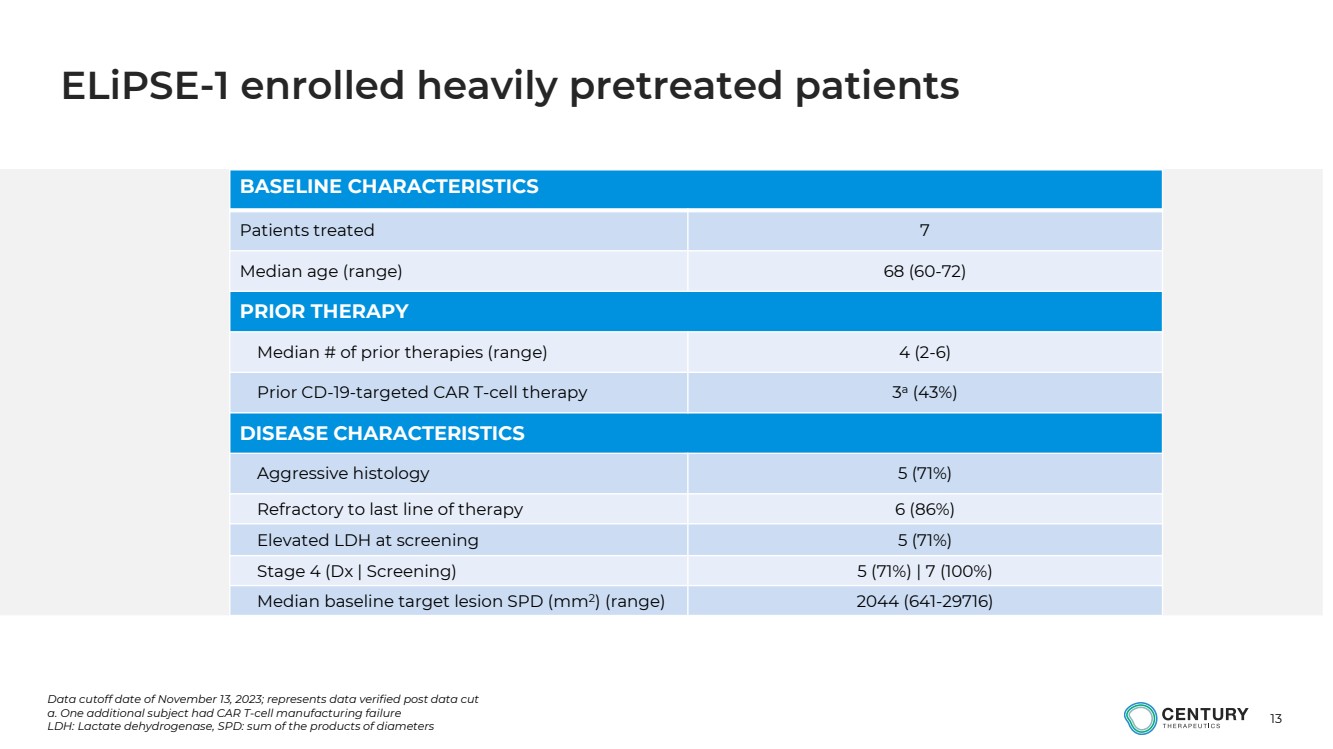
| 13
BASELINE CHARACTERISTICS
Patients treated 7
Median age (range) 68 (60-72)
PRIOR THERAPY
Median # of prior therapies (range) 4 (2-6)
Prior CD-19-targeted CAR T-cell therapy 3
a
(43%)
DISEASE CHARACTERISTICS
Aggressive histology 5 (71%)
Refractory to last line of therapy 6 (86%)
Elevated LDH at screening 5 (71%)
Stage 4 (Dx | Screening) 5 (71%) | 7 (100%)
Median baseline target lesion SPD (mm2
) (range) 2044 (641-29716)
Data cutoff date of November 13, 2023; represents data verified post data cut
a. One additional subject had CAR T-cell manufacturing failure
LDH: Lactate dehydrogenase, SPD: sum of the products of diameters
ELiPSE-1 enrolled heavily pretreated patients |
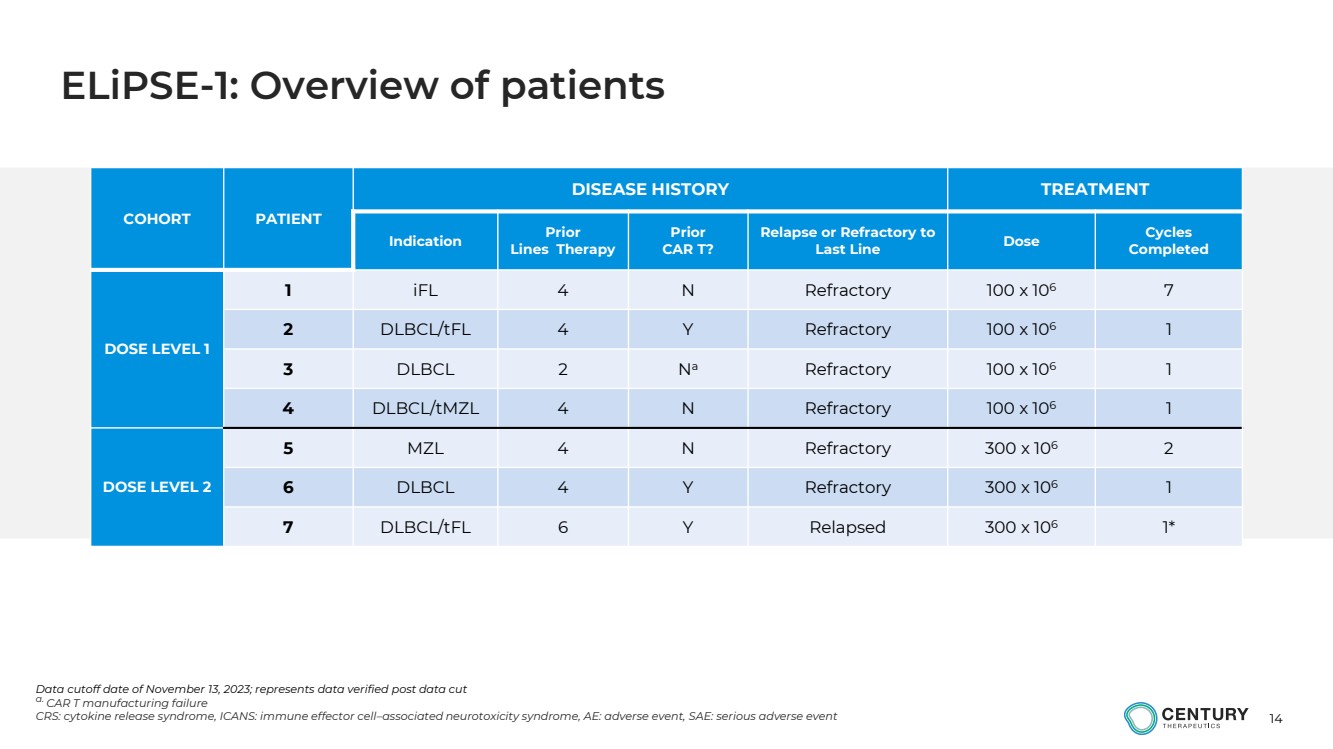
| 14
COHORT PATIENT
DISEASE HISTORY TREATMENT
Indication Prior
Lines Therapy
Prior
CAR T?
Relapse or Refractory to
Last Line Dose Cycles
Completed
DOSE LEVEL 1
1 iFL 4 N Refractory 100 x 106 7
2 DLBCL/tFL 4 Y Refractory 100 x 106 1
3 DLBCL 2 Na Refractory 100 x 106 1
4 DLBCL/tMZL 4 N Refractory 100 x 106 1
DOSE LEVEL 2
5 MZL 4 N Refractory 300 x 106 2
6 DLBCL 4 Y Refractory 300 x 106 1
7 DLBCL/tFL 6 Y Relapsed 300 x 106 1*
Data cutoff date of November 13, 2023; represents data verified post data cut
a. CAR T manufacturing failure
CRS: cytokine release syndrome, ICANS: immune effector cell–associated neurotoxicity syndrome, AE: adverse event, SAE: serious adverse event
ELiPSE-1: Overview of patients |
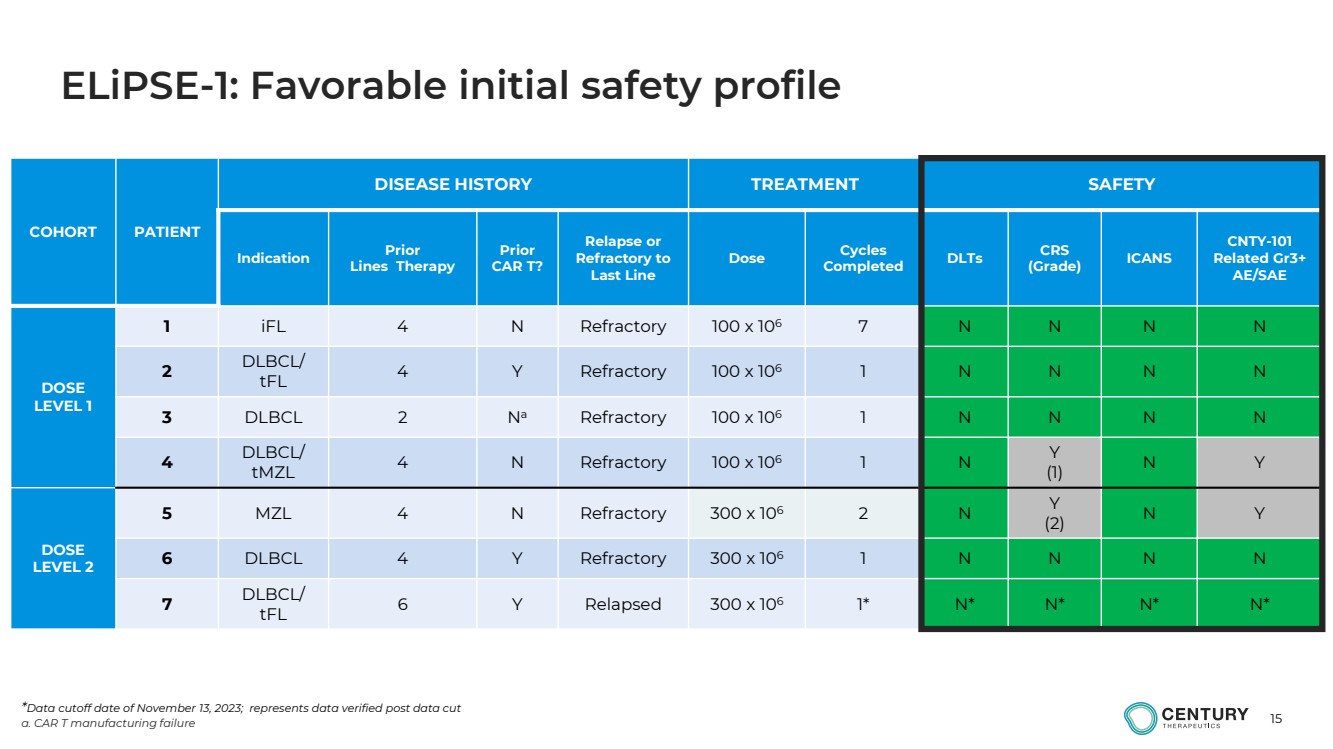
| 15
COHORT PATIENT
DISEASE HISTORY TREATMENT SAFETY
Indication Prior
Lines Therapy
Prior
CAR T?
Relapse or
Refractory to
Last Line
Dose Cycles
Completed DLTs CRS
(Grade) ICANS
CNTY-101
Related Gr3+
AE/SAE
DOSE
LEVEL 1
1 iFL 4 N Refractory 100 x 106 7 N N N N
2
DLBCL/
tFL 4 Y Refractory 100 x 106 1 N N N N
3 DLBCL 2 Na Refractory 100 x 106 1 N N N N
4
DLBCL/
tMZL 4 N Refractory 100 x 106 1 N
Y
(1) N Y
DOSE
LEVEL 2
5 MZL 4 N Refractory 300 x 106 2 N
Y
(2) N Y
6 DLBCL 4 Y Refractory 300 x 106 1 N N N N
7
DLBCL/
tFL 6 Y Relapsed 300 x 106 1* N* N* N* N*
*Data cutoff date of November 13, 2023; represents data verified post data cut
a. CAR T manufacturing failure
ELiPSE-1: Favorable initial safety profile |
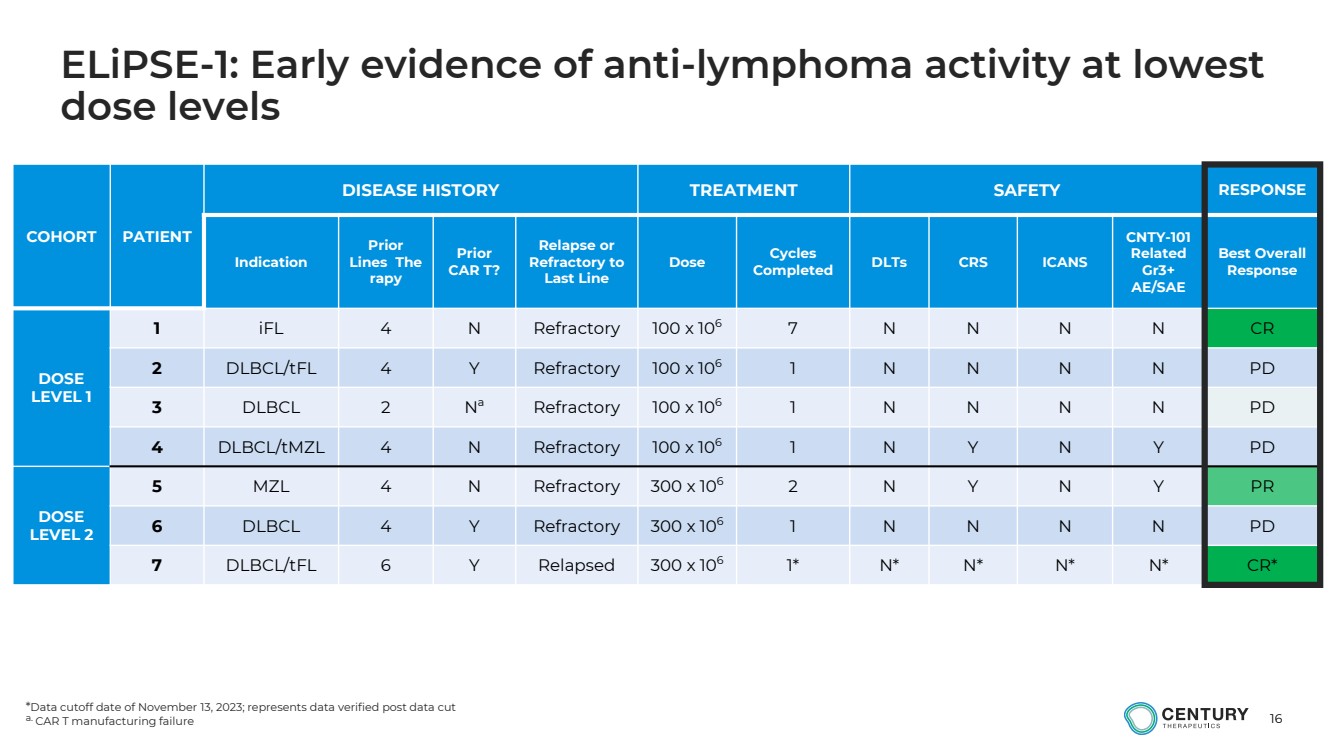
| 16
ELiPSE-1: Early evidence of anti-lymphoma activity at lowest
dose levels
COHORT PATIENT
DISEASE HISTORY TREATMENT SAFETY RESPONSE
Indication
Prior
Lines The
rapy
Prior
CAR T?
Relapse or
Refractory to
Last Line
Dose Cycles
Completed DLTs CRS ICANS
CNTY-101
Related
Gr3+
AE/SAE
Best Overall
Response
DOSE
LEVEL 1
1 iFL 4 N Refractory 100 x 106 7 N N N N CR
2 DLBCL/tFL 4 Y Refractory 100 x 106
1 N N N N PD
3 DLBCL 2 N
a Refractory 100 x 106
1 N N N N PD
4 DLBCL/tMZL 4 N Refractory 100 x 106
1 N Y N Y PD
DOSE
LEVEL 2
5 MZL 4 N Refractory 300 x 106 2 N Y N Y PR
6 DLBCL 4 Y Refractory 300 x 106
1 N N N N PD
7 DLBCL/tFL 6 Y Relapsed 300 x 106
1* N* N* N* N* CR*
*Data cutoff date of November 13, 2023; represents data verified post data cut
a. CAR T manufacturing failure |
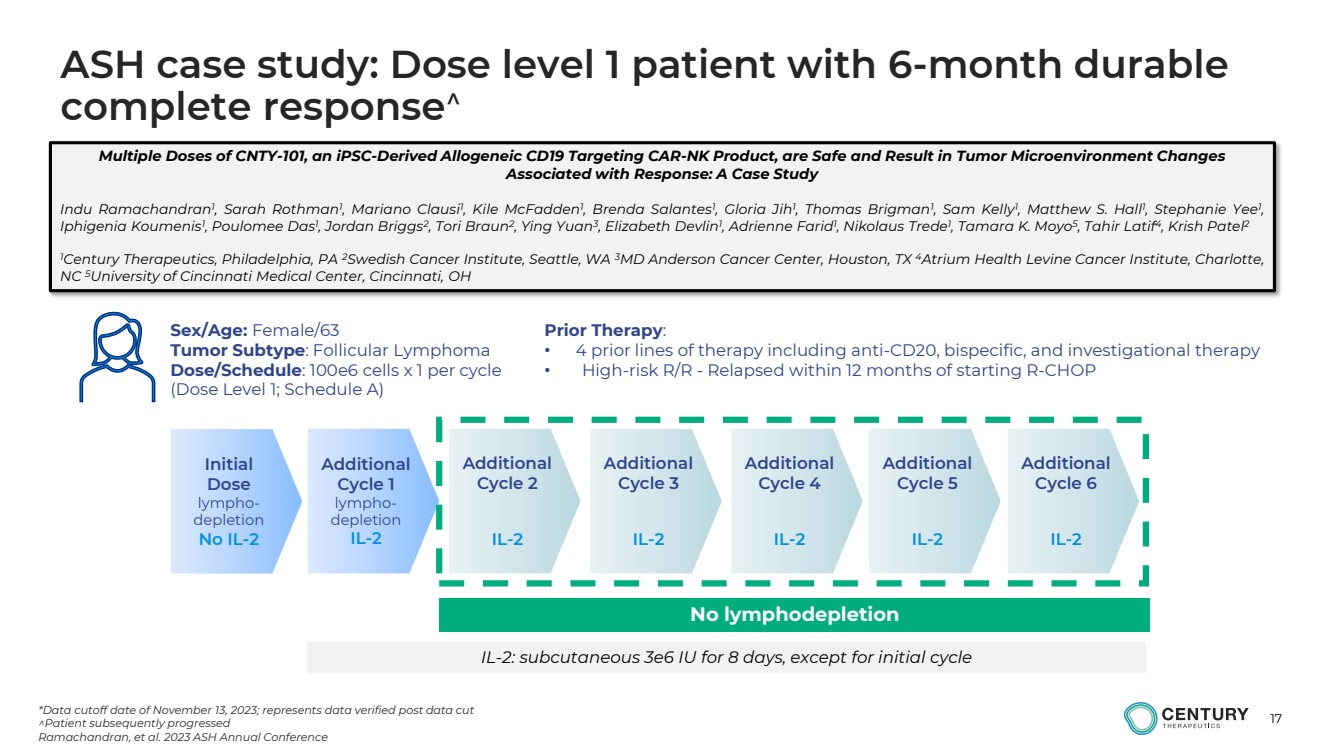
| 17
Initial
Dose
lympho-depletion
No IL-2
Sex/Age: Female/63
Tumor Subtype: Follicular Lymphoma
Dose/Schedule: 100e6 cells x 1 per cycle
(Dose Level 1; Schedule A)
Prior Therapy:
• 4 prior lines of therapy including anti-CD20, bispecific, and investigational therapy
• High-risk R/R - Relapsed within 12 months of starting R-CHOP
Additional
Cycle 1
lympho-depletion
IL-2
Additional
Cycle 2
IL-2
Additional
Cycle 3
IL-2
Additional
Cycle 4
IL-2
Additional
Cycle 5
IL-2
Additional
Cycle 6
IL-2
*Data cutoff date of November 13, 2023; represents data verified post data cut
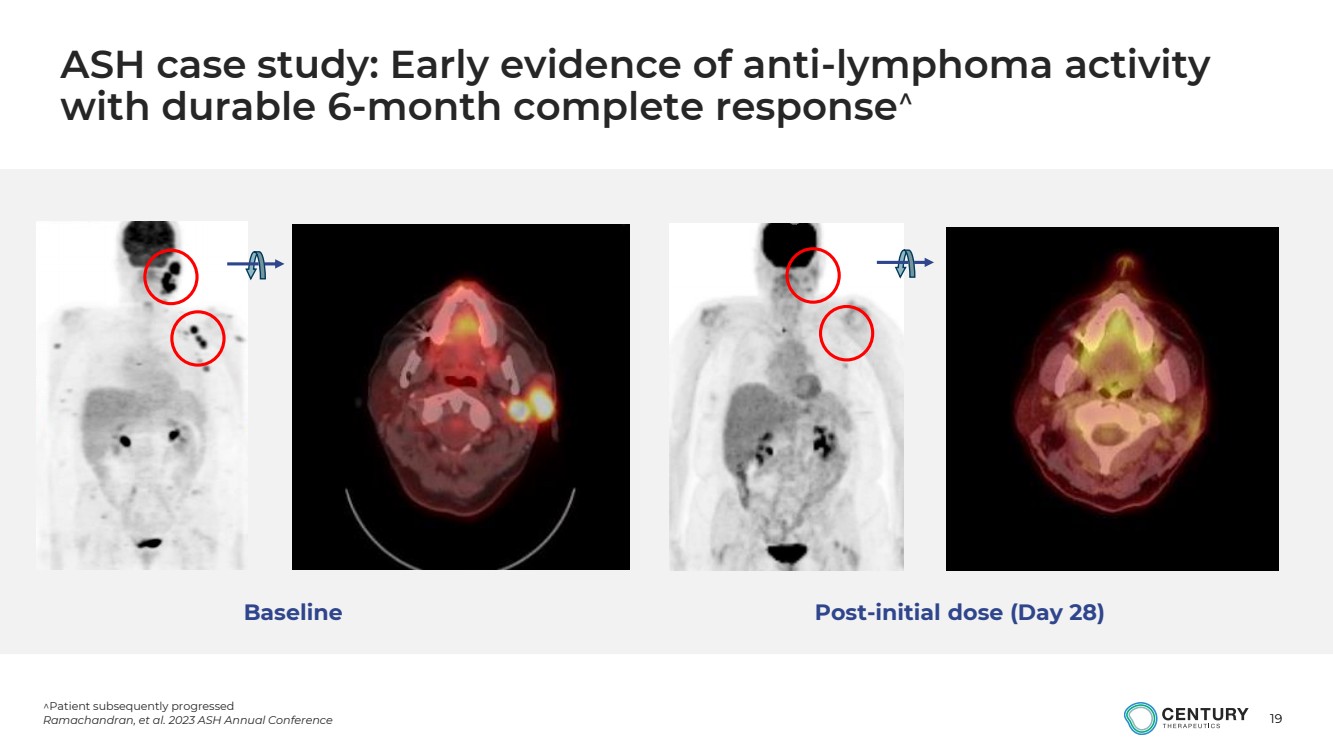
Patient subsequently progressed
Ramachandran, et al. 2023 ASH Annual Conference
IL-2: subcutaneous 3e6 IU for 8 days, except for initial cycle
No lymphodepletion
Multiple Doses of CNTY-101, an iPSC-Derived Allogeneic CD19 Targeting CAR-NK Product, are Safe and Result in Tumor Microenvironment Changes
Associated with Response: A Case Study
Indu Ramachandran1
, Sarah Rothman1
, Mariano Clausi1
, Kile McFadden1
, Brenda Salantes1
, Gloria Jih1
, Thomas Brigman1
, Sam Kelly1
, Matthew S. Hall1
, Stephanie Yee1
,
Iphigenia Koumenis1
, Poulomee Das1
, Jordan Briggs2
, Tori Braun2
, Ying Yuan3
, Elizabeth Devlin1
, Adrienne Farid1
, Nikolaus Trede1
, Tamara K. Moyo5
, Tahir Latif4, Krish Patel2
1Century Therapeutics, Philadelphia, PA 2Swedish Cancer Institute, Seattle, WA 3MD Anderson Cancer Center, Houston, TX 4Atrium Health Levine Cancer Institute, Charlotte,
NC 5University of Cincinnati Medical Center, Cincinnati, OH
ASH case study: Dose level 1 patient with 6-month durable
complete response |
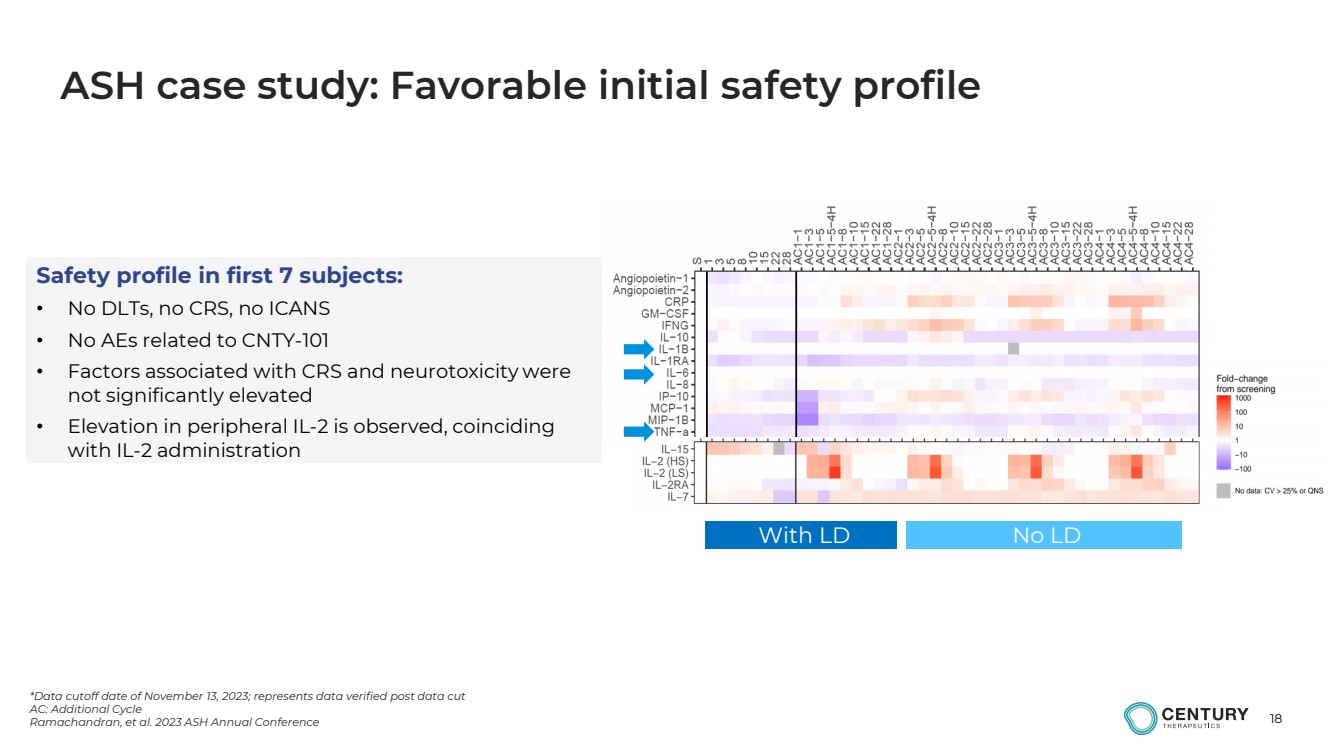
| 18
*Data cutoff date of November 13, 2023; represents data verified post data cut
AC: Additional Cycle
Ramachandran, et al. 2023 ASH Annual Conference
ASH case study: Favorable initial safety profile
With LD No LD
Safety profile in first 7 subjects:
• No DLTs, no CRS, no ICANS
• No AEs related to CNTY-101
• Factors associated with CRS and neurotoxicity were
not significantly elevated
• Elevation in peripheral IL-2 is observed, coinciding
with IL-2 administration |
| 19
Patient subsequently progressed
Ramachandran, et al. 2023 ASH Annual Conference
ASH case study: Early evidence of anti-lymphoma activity
with durable 6-month complete response
Baseline Post-initial dose (Day 28) |
| 20
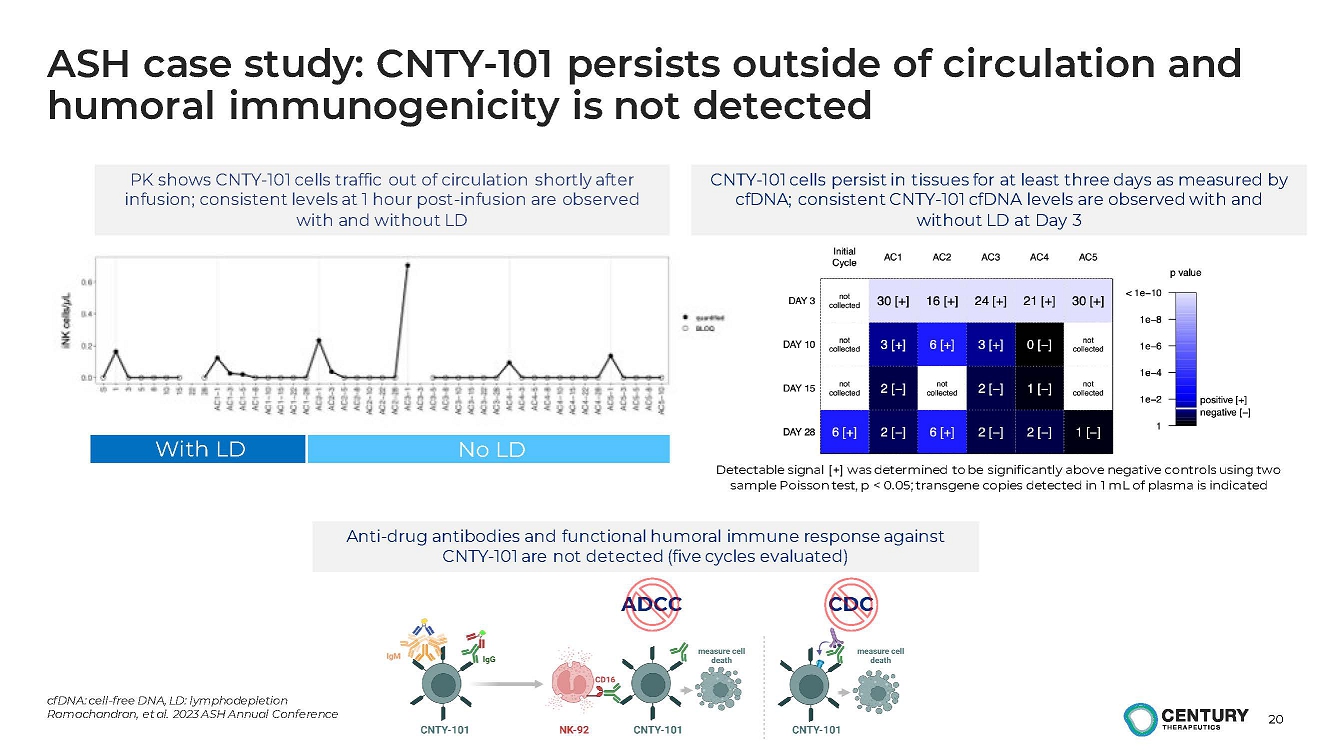
ASH case study: CNTY-101 persists outside of circulation and
humoral immunogenicity is not detected
cfDNA: cell-free DNA, LD: lymphodepletion
Ramachandran, et al. 2023 ASH Annual Conference
PK shows CNTY-101 cells traffic out of circulation shortly after
infusion; consistent levels at 1 hour post-infusion are observed
with and without LD
Detectable signal [+] was determined to be significantly above negative controls using two
sample Poisson test, p < 0.05; transgene copies detected in 1 mL of plasma is indicated
CNTY-101 cells persist in tissues for at least three days as measured by
cfDNA; consistent CNTY-101 cfDNA levels are observed with and
without LD at Day 3.
With LD No LD
Anti-drug antibodies and functional humoral immune response against
CNTY-101 are not detected (five cycles evaluated).
ADCC CDC |
| 21
ASH case study: Intra-tumoral adaptive immune response
observed following initial dose without IL-2
CD8Ki67 CD8Ki67
CD56Ki67 CD56Ki67
CARTNFAIFNG CARTNFAIFNG
Total Ki67+
TNF-α IFN-γ
Total Ki67+
0
200
400
600
800
1000 Cells per mm2 CD8+
T cells
Endogenous
NK cells
Secreting
cells
SCREEN DAY 8 |

| 22
Summary of ELiPSE-1 data
• Heavily pretreated and refractory patient population treated in first-in-human dose escalation trial
• Favorable safety profile; can be delivered in an outpatient setting
• Encouraging early efficacy signals at lowest dose levels
o 2 patients achieving CR, including 1 patient with 6-month durable CR
• No evidence of allo-rejection
• Initial data for CNTY-101 supports the potential for Allo-Evasion™ to enable a multi-dosing regimen without the need for continued lymphodepletion
• We believe CNTY-101’s manageable initial safety profile, initial response data, and PK/PD
supports advancing to higher doses to potentially deepen and prolong clinical response
Cohorts of 1 billion cells/1 monthly dose and 300 million/weekly x 3 doses are open;
Additional clinical data expected in mid-2024 |

| CNTY-101 in Systemic Lupus
Erythematosus |

| 24
Estimated global prevalence of 3.4
million patients1
• Abnormal B cell function and
autoantibody production are central to
disease pathogenesis
• Major causes of morbidity and mortality
involve multiple systems
o Renal, CNS and cardiovascular
involvement are major causes of
morbidity and mortality
Despite approved treatments, significant unmet need remains
• Chronic treatment with broad-acting anti-inflammatory and
immunosuppressives
• Current treatments fail to significantly impact morbidity in the
moderate to severe population
• Treatment toxicity and disease flares remain common
Autologous anti-CD19 CAR T cell therapies have established a
promising efficacy proof of concept in SLE2
• Challenges remain due to potential exposure to CRS and ICANS, product
availability, and long-term risks including B-cell aplasia
Opportunity in systemic lupus erythematosus to improve
long-term disease control
1. Tian J, et al. Ann Rheum Dis 2023;82:351–356 http://dx.doi.org/10.1136/ard-2022-223035
2. Mackensen A, et al.. Nature Medicine 2022 28:10 (2124-2132) https://doi.org/10.1038/s41591-022-02017-5
CNS: central nervous system, SLE: systemic lupus erythematosus |

| 25
CNTY-101 aims to eliminate pathogenic B-cells in SLE leading
to remission via repeat dosing facilitated by Allo-EvasionTM
CNTY-101 has the potential to improve on current SLE treatments
• Anti-CD19 CAR-iNK cells derived from an HDR precision-edited iPSC clone, including IL-15 cytokine
support, a safety switch, and Allo-EvasionTM edits
• Clonal, consistent, well-characterized product
• Available off-the-shelf, without requiring patient apheresis, no manufacturing wait time
• Favorable initial safety profile, allowing for outpatient treatment
• Ability to be redosed without lymphodepletion, while avoiding allo-rejection based on initial data
• Potential to enable B cell depletion and a reduction in auto-antibodies without prolonged B-cell aplasia
Aim: Safely provide immune reset with an immediately available therapy
HDR: homology-directed repair |
| 26
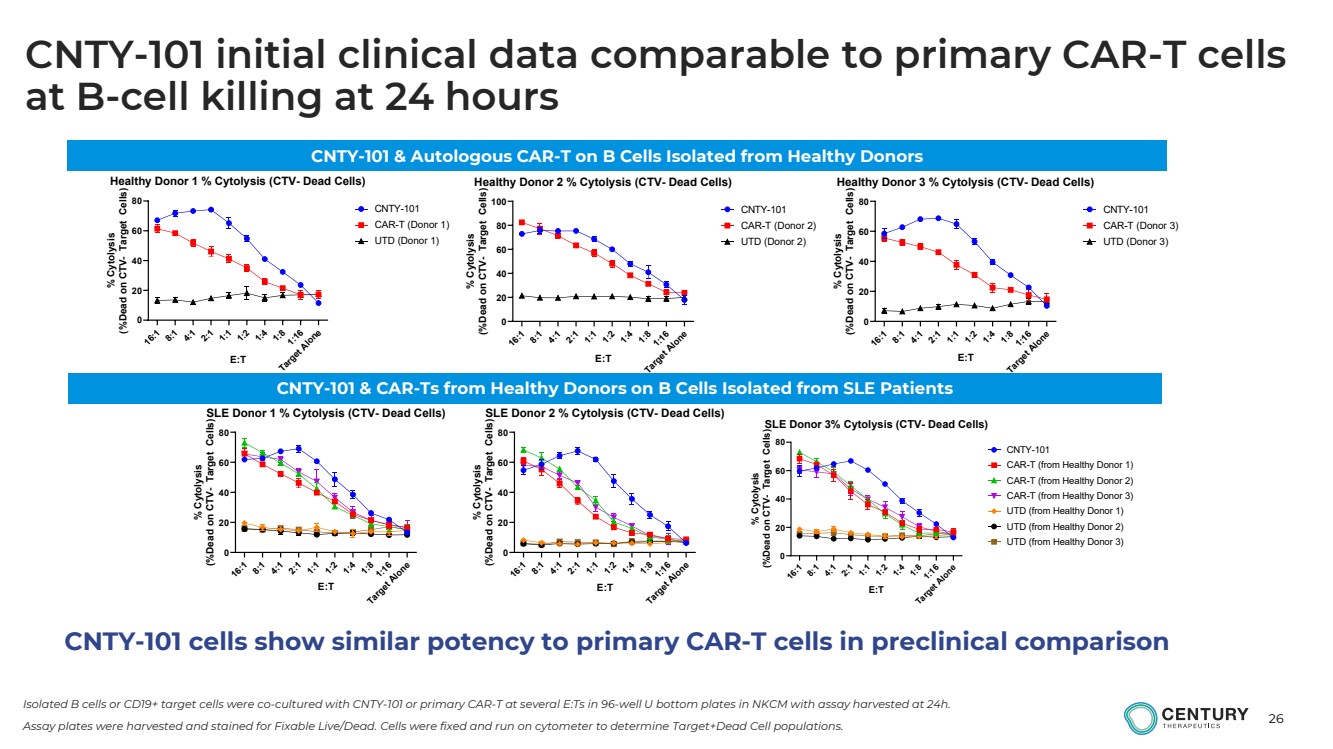
Isolated B cells or CD19+ target cells were co-cultured with CNTY-101 or primary CAR-T at several E:Ts in 96-well U bottom plates in NKCM with assay harvested at 24h.
Assay plates were harvested and stained for Fixable Live/Dead. Cells were fixed and run on cytometer to determine Target+Dead Cell populations. 16:1
8:1
4:1
2:1
1:1
1:2
1:4
1:8
1:16 Target Alone
0
20
40
60
80 Healthy Donor 1 % Cytolysis (CTV- Dead Cells)
E:T
% Cytolysis
(%Dead on CTV- Target Cells)
CNTY-101 CAR-T (Donor 1) UTD (Donor 1)
16:1
8:1
4:1
2:1
1:1
1:2
1:4
1:8
1:16 Target Alone
0
20
40
60
80
100 Healthy Donor 2 % Cytolysis (CTV- Dead Cells)
E:T
% Cytolysis
(%Dead on CTV- Target Cells)
CNTY-101 CAR-T (Donor 2) UTD (Donor 2)
16:1
8:1
4:1
2:1
1:1
1:2
1:4
1:8
1:16 Target Alone
0
20
40
60
80 Healthy Donor 3 % Cytolysis (CTV- Dead Cells)
E:T
% Cytolysis
(%Dead on CTV- Target Cells)
CNTY-101 CAR-T (Donor 3) UTD (Donor 3)
16:1
8:1
4:1
2:1
1:1
1:2
1:4
1:8
1:16 Target Alone
0
20
40
60
80 SLE Donor 1 % Cytolysis (CTV- Dead Cells)
E:T
% Cytolysis
(%Dead on CTV- Target Cells)
16:1
8:1
4:1
2:1
1:1
1:2
1:4
1:8
1:16 Target Alone
0
20
40
60
80 SLE Donor 2 % Cytolysis (CTV- Dead Cells)
E:T
% Cytolysis
(%Dead on CTV- Target Cells)
16:1
8:1
4:1
2:1
1:1
1:2
1:4
1:8
1:16 Target Alone
0
20
40
60
80 SLE Donor 3% Cytolysis (CTV- Dead Cells)
E:T
% Cytolysis
(%Dead on CTV- Target Cells)
CNTY-101 CAR-T (from Healthy Donor 1) CAR-T (from Healthy Donor 2) CAR-T (from Healthy Donor 3) UTD (from Healthy Donor 1) UTD (from Healthy Donor 2) UTD (from Healthy Donor 3)
CNTY-101 & Autologous CAR-T on B Cells Isolated from Healthy Donors
CNTY-101 & CAR-Ts from Healthy Donors on B Cells Isolated from SLE Patients
CNTY-101 cells show similar potency to primary CAR-T cells in preclinical comparison
CNTY-101 initial clinical data comparable to primary CAR-T cells
at B-cell killing at 24 hours |
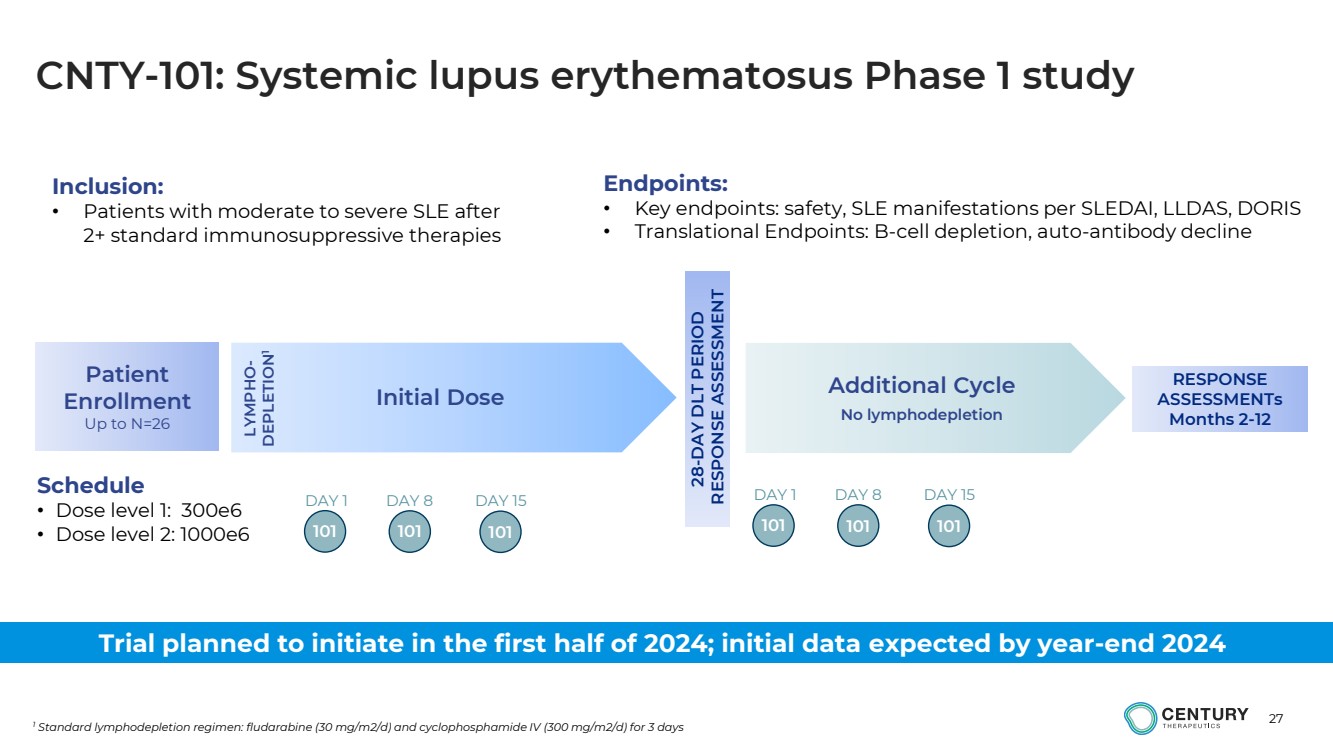
| 27
CNTY-101: Systemic lupus erythematosus Phase 1 study
1 Standard lymphodepletion regimen: fludarabine (30 mg/m2/d) and cyclophosphamide IV (300 mg/m2/d) for 3 days
Initial Dose Additional Cycle
No lymphodepletion
Schedule
• Dose level 1: 300e6
• Dose level 2: 1000e6 LYMPHO-DEPLETION1
28-DAY DLT PERIOD
RESPONSE ASSESSMENT
Patient
Enrollment
Up to N=26
101
DAY 1 DAY 8
101 101
DAY 15
101
DAY 1 DAY 8
101 101
DAY 15
Inclusion:
• Patients with moderate to severe SLE after
2+ standard immunosuppressive therapies
RESPONSE
ASSESSMENTs
Months 2-12
Endpoints:
• Key endpoints: safety, SLE manifestations per SLEDAI, LLDAS, DORIS
• Translational Endpoints: B-cell depletion, auto-antibody decline
Trial planned to initiate in the first half of 2024; initial data expected by year-end 2024 |
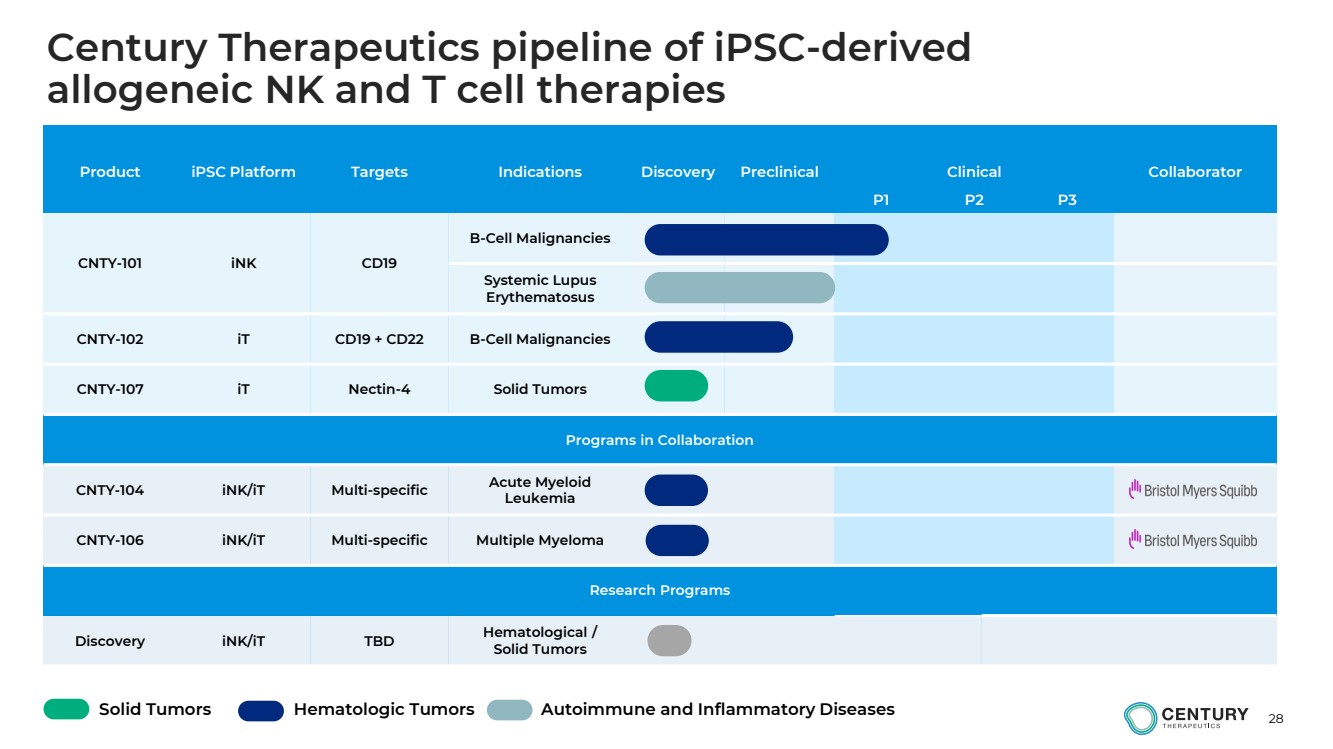
| 28
Product iPSC Platform Targets Indications Discovery Preclinical Clinical Collaborator
P1 P2 P3
CNTY-101 iNK CD19
B-Cell Malignancies
Systemic Lupus
Erythematosus
CNTY-102 iT CD19 + CD22 B-Cell Malignancies
CNTY-107 iT Nectin-4 Solid Tumors
Programs in Collaboration
CNTY-104 iNK/iT Multi-specific Acute Myeloid
Leukemia
CNTY-106 iNK/iT Multi-specific Multiple Myeloma
Research Programs
Discovery iNK/iT TBD Hematological /
Solid Tumors
Solid Tumors Hematologic Tumors Autoimmune and Inflammatory Diseases
Century Therapeutics pipeline of iPSC-derived
allogeneic NK and T cell therapies |

| Closing |

| 30
Century Therapeutics: Building an industry-leading, next-generation allogeneic iPSC-derived cell therapy platform
LIMITLESS POTENTIAL...
PRECISION DESIGN...
ENDURING IMPACT...
Foundational investments in iPSC technology,
genetic editing, and manufacturing
Progressing multiple clinical programs in
oncology and autoimmune and inflammatory
diseases
Well-capitalized into 2026 to enable delivery
on key milestones and clinical data |

| 31
Q&A participants
• Brent Pfeiffenberger, Pharm.D., Chief Executive Officer
• Hy Levitsky, M.D., President of Research and Development
• Nick Trede, M.D., Ph.D., SVP, Head of Clinical Development
• Adrienne Farid, Ph.D., Chief Operations Officer and Head of Early Development
• Michael Diem, M.D., Chief Financial Officer
• Greg Russotti, Ph.D., Chief Technology and Manufacturing Officer |
| Initial Data from Phase 1 ELiPSE-1 Trial
of CNTY-101 in Relapsed/Refractory B-cell Lymphomas and Overview of
Planned Phase 1 Study in Systemic
Lupus Erythematosus
December 11, 2023 |
Cover
|
Dec. 09, 2023 |
| Cover [Abstract] |
|
| Document Type |
8-K
|
| Amendment Flag |
false
|
| Document Period End Date |
Dec. 09, 2023
|
| Entity File Number |
001-40498
|
| Entity Registrant Name |
Century Therapeutics, Inc.
|
| Entity Central Index Key |
0001850119
|
| Entity Tax Identification Number |
84-2040295
|
| Entity Incorporation, State or Country Code |
DE
|
| Entity Address, Address Line One |
25
North 38th Street
|
| Entity Address, Address Line Two |
11th Floor
|
| Entity Address, City or Town |
Philadelphia
|
| Entity Address, State or Province |
PA
|
| Entity Address, Postal Zip Code |
19104
|
| City Area Code |
267
|
| Local Phone Number |
817-5790
|
| Written Communications |
false
|
| Soliciting Material |
false
|
| Pre-commencement Tender Offer |
false
|
| Pre-commencement Issuer Tender Offer |
false
|
| Title of 12(b) Security |
Common Stock, par value $0.0001 per share
|
| Trading Symbol |
IPSC
|
| Security Exchange Name |
NASDAQ
|
| Entity Emerging Growth Company |
true
|
| Elected Not To Use the Extended Transition Period |
false
|
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 2 such as Street or Suite number
| Name: |
dei_EntityAddressAddressLine2 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the state or province.
| Name: |
dei_EntityAddressStateOrProvince |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:stateOrProvinceItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCommission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.
| Name: |
dei_EntityFileNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fileNumberItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 13e
-Subsection 4c
| Name: |
dei_PreCommencementIssuerTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 14d
-Subsection 2b
| Name: |
dei_PreCommencementTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTitle of a 12(b) registered security. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b
| Name: |
dei_Security12bTitle |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:securityTitleItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the Exchange on which a security is registered. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection d1-1
| Name: |
dei_SecurityExchangeName |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarExchangeCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Section 14a
-Number 240
-Subsection 12
| Name: |
dei_SolicitingMaterial |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:tradingSymbolItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Securities Act
-Number 230
-Section 425
| Name: |
dei_WrittenCommunications |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
Grafico Azioni Century Therapeutics (NASDAQ:IPSC)
Storico
Da Apr 2024 a Mag 2024

Grafico Azioni Century Therapeutics (NASDAQ:IPSC)
Storico
Da Mag 2023 a Mag 2024
