LONDON, March 8,
2023 /PRNewswire/ -- Smith+Nephew (LSE:SN)
(NYSE:SNN), the global medical technology company, today announces
it will showcase its latest Sports Medicine procedural innovations
for joint repair and arthroscopic enabling technologies during the
American Academy of Orthopaedic Surgeons 2023 Annual Meeting being
held in Las Vegas, NV. These
include:
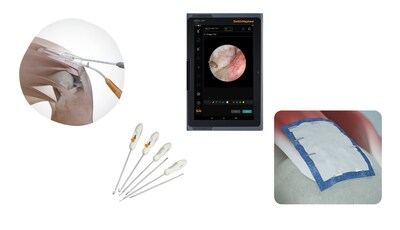
UltraTRAC™ QUAD ACL Reconstruction Technique: New
technologies combine to deliver an advanced, comprehensive
procedural solution for surgeons who want to expand their ligament
graft options and maximize the variety of patients they can
accommodate. Together, the new QUADTRAC™ Quadriceps Tendon
Harvest Guide System, X-WING™ Graft Preparation System and
ULTRABUTTON™ Family of Adjustable Fixation Devices provide a
controlled and reproducible technique to harvest only the desired
quadriceps tendon tissue.1-3 Quadriceps ACL
reconstruction is a growing procedure that offers many advantages
including a lower risk of anterior knee pain4-6 and
predictable, larger graft diameter4,7,8 compared to
bone-tendon-bone or hamstring autograft options respectively.
INTELLIO™ Tablet: Helping streamline your procedures
with simple, centralized connectivity in the palm of your hand -
the INTELLIO Tablet is a medical-grade device designed for use with
Smith+Nephew's INTELLIO Connected Tower. Supporting both wireless
and tethered connectivity, the INTELLIO Tablet increases users'
ability to control and adjust the LENS 4K Surgical Imaging System, DYONICS™ POWER
II Control System, WEREWOLF™ COBLATION™ System and the
DOUBLEFLO™ Inflow/Outflow Pump.
CAP-FIX™ Capsular Management Family: A comprehensive
range of products designed to maximize the efficiency of capsular
management procedures. The CAP-FIX Suture Passer reduces the number
of steps required for capsule closure, compared to suture
shuttling,9 and is optimized for sharpness, strength,
trajectory and suture throw.9-10 CAP-FIX Blades are
offered as curved or straight with a choice of a pencil grip or a
handle for increased leverage. Each blade has a consistent
sharpness to cut through hip capsular tissue.11 From
open to close, the CAP-FIX Capsular Management Family and the full
portfolio of hip preservation products encourages surgeons to "Go
Beyond the Repair."
REGENETEN™ Bioinductive Implant: Supports the
body's natural healing response to facilitate new tendon-like
tissue growth and changes the course of rotator cuff tear
progression.12-17 The implant is about the size of a
postage stamp and has been shown to be completely resorbed within
six months.*14,17 In addition to rotator cuff repair,
the REGENETEN Bioinductive Implant can be used for hip repair as
well as Achilles repair and reconstruction.
To learn more about these ground-breaking Sports Medicine
innovations, please stop by the Smith+Nephew booth (#2021) during
AAOS 2023
References
*On human biopsy (n=1) and in-vivo sampling
1 Smith+Nephew 2022. QUADTRAC Marketing Claims.
Internal Report. 15011572 Rev A.
2 Smith+Nephew 2022.TRAC-Cutter Push-Pull Cut Force DV
Protocol. Internal Report. 15011552 Rev A.
3 Smith+Nephew 2022.QUAD-Cutter Force to Cut Design
Verification. Internal Report. 15011559 Rev A.
4 Buerba RA, Boden SA, Lesniak B. Graft Selection in
Contemporary Anterior Cruciate Ligament Reconstruction. JAAOS:
Global Research and Reviews. 2021;5(10).
5 Lund, Bent et al. "Is
Quadriceps Tendon a Better Graft Choice Than Patellar Tendon? A
Prospective Randomized Study." Arthroscopy 2014; 30(5):
593-598..
6 Malinowski K, Paszkowski J, Mostowy M, Goralczyk A,
Laprade RF, Hermanowicz K. Quadriceps Tendon-Bone Full-Thickness
Autograft: Reproducible and Easy Harvesting Technique Using Simple
Surgical Tools. Arthroscopy Techniques. 2021;10(4).
7 Todor A, Nistor DV, Caterev S. Clinical outcomes after
ACL reconstruction with free quadriceps tendon autograft versus
hamstring tendons autograft. A retrospective study with a minimal
follow-up two years. Acta Orthop Traumatol Turc.
2019;53(3):180-183.
8 Runer A, Csapo R, Hepperger C, Herbort M, Hoser C,
Fink C. Anterior Cruciate Ligament Reconstructions With Quadriceps
Tendon Autograft Result in Lower Graft Rupture Rates but Similar
Patient-Reported Outcomes as Compared With Hamstring Tendon
Autograft: A Comparison of 875 Patients. AJSM. 2020;48(9).
9 Smith+Nephew 2021. Internal report 15011184
A.
10 Smith+Nephew 2021. Internal report 15011185 A.
11 Smith+Nephew 2020. CAP-FIX BLADE Claims Testing.
Internal Report. 15009423 B.
12 Bokor DJ, Sonnabend D, Deady L, et al. Evidence of
healing of partial-thickness rotator cuff tears following
arthroscopic augmentation with a collagen implant: a 2-year MRI
follow-up. Muscles, Ligaments Tendons J. 2016;6(1):16-25.
13 Schlegel TF, Abrams JS, Bushnell BD, Brock JL, Ho CP.
Radiologic and clinical evaluation of a bioabsorbable collagen
implant to treat partial-thickness tears: a prospective multicenter
study. J Shoulder Elbow Surg. 2018 27(2):242-251.
14 Van Kampen C, Arnoczky
S, Parks P, et al. Tissue-engineered augmentation of a rotator cuff
tendon using a reconstituted collagen scaffold: a histological
evaluation in sheep. Muscles Ligaments Tendons J.
2013;3(3):229-235.
15 Bokor DJ, Sonnabend DH, Deady L, et al. Healing of
partial-thickness rotator cuff tears following arthroscopic
augmentation with a highly porous collagen implant: a 5-year
clinical and MRI follow-up. Muscles, Ligaments Tendons J.
2019;9(3):338-347.
16 McElvany MD, McGoldrick E, Gee AO, Neradilek MB,
Matsen FA, 3rd. Rotator cuff repair: published evidence on factors
associated with repair integrity and clinical outcome. Am J Sports
Med. 2015;43(2):491-500.
17 Arnoczky SP, Bishai SK, Schofield B, et al.
Histologic Evaluation of Biopsy Specimens Obtained After Rotator
Cuff Repair Augmented With a Highly Porous Collagen Implant.
Arthroscopy. 2017;33(2):278-283
About Smith+Nephew
Smith+Nephew is a portfolio medical
technology company focused on the repair, regeneration and
replacement of soft and hard tissue. We exist to restore people's
bodies and their self-belief by using technology to take the limits
off living. We call this purpose 'Life Unlimited'. Our 19,000
employees deliver this mission every day, making a difference to
patients' lives through the excellence of our product
portfolio, and the invention and application of new technologies
across our three global franchises of Orthopaedics, Sports
Medicine & ENT and Advanced Wound Management.
Founded in Hull, UK, in 1856,
we now operate in more than 100 countries, and generated annual
sales of $5.2 billion in 2022.
Smith+Nephew is a constituent of the FTSE100 (LSE:SN, NYSE:SNN).
The terms 'Group' and 'Smith+Nephew' are used to refer to Smith
& Nephew plc and its consolidated subsidiaries, unless the
context requires otherwise.
For more information about Smith+Nephew, please
visit www.smith-nephew.com and follow us
on Twitter, LinkedIn, Instagram or Facebook.
Forward-looking Statements
This document may contain forward-looking statements that may
or may not prove accurate. For example, statements regarding
expected revenue growth and trading margins, market trends and our
product pipeline are forward-looking statements. Phrases such as
"aim", "plan", "intend", "anticipate", "well-placed", "believe",
"estimate", "expect", "target", "consider" and similar expressions
are generally intended to identify forward-looking statements.
Forward-looking statements involve known and unknown risks,
uncertainties and other important factors that could cause actual
results to differ materially from what is expressed or implied by
the statements. For Smith+Nephew, these factors include: risks
related to the impact of COVID-19, such as the depth and longevity
of its impact, government actions and other restrictive measures
taken in response, material delays and cancellations of elective
procedures, reduced procedure capacity at medical facilities,
restricted access for sales representatives to medical facilities,
or our ability to execute business continuity plans as a result of
COVID-19; economic and financial conditions in the markets we
serve, especially those affecting health care providers, payers and
customers (including, without limitation, as a result of COVID-19);
price levels for established and innovative medical devices;
developments in medical technology; regulatory approvals,
reimbursement decisions or other government actions; product
defects or recalls or other problems with quality management
systems or failure to comply with related regulations; litigation
relating to patent or other claims; legal compliance risks and
related investigative, remedial or enforcement actions; disruption
to our supply chain or operations or those of our suppliers
(including, without limitation, as a result of COVID-19);
competition for qualified personnel; strategic actions, including
acquisitions and dispositions, our success in performing due
diligence, valuing and integrating acquired businesses; disruption
that may result from transactions or other changes we make in our
business plans or organisation to adapt to market developments; and
numerous other matters that affect us or our markets, including
those of a political, economic, business, competitive or
reputational nature. Please refer to the documents that
Smith+Nephew has filed with the U.S. Securities and Exchange
Commission under the U.S. Securities Exchange Act of 1934, as
amended, including Smith+Nephew's most recent annual report on Form
20-F, for a discussion of certain of these factors. Any
forward-looking statement is based on information available to
Smith+Nephew as of the date of the statement. All written or oral
forward-looking statements attributable to Smith+Nephew are
qualified by this caution. Smith+Nephew does not undertake any
obligation to update or revise any forward-looking statement to
reflect any change in circumstances or in Smith+Nephew's
expectations.
™ Trademark of Smith+Nephew. Certain marks
registered US Patent and Trademark Office.
 View original content to download
multimedia:https://www.prnewswire.com/news-releases/smithnephew-to-highlight-procedural-innovations-in-sports-medicine-during-aaos-2023-annual-meeting-301764101.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/smithnephew-to-highlight-procedural-innovations-in-sports-medicine-during-aaos-2023-annual-meeting-301764101.html
SOURCE Smith & Nephew plc