0001850119
false
0001850119
2023-08-09
2023-08-09
iso4217:USD
xbrli:shares
iso4217:USD
xbrli:shares
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, DC 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported):
August 9, 2023
Century Therapeutics, Inc.
(Exact name of registrant as specified in its charter)
| Delaware |
|
001-40498 |
|
84-2040295 |
(State or other jurisdiction of
incorporation or organization) |
|
(Commission File Number) |
|
(I.R.S. Employer
Identification No.) |
|
25
North 8th Street, 11th Floor
Philadelphia, Pennsylvania |
|
19104 |
| (Address of principal executive offices) |
|
(Zip Code) |
Registrant’s telephone number, including
area code: (267) 817-5790
Not Applicable
(Former name or former address, if changed since
last report)
Check the appropriate box below if the Form 8-K filing is intended
to simultaneously satisfy the filing obligation of the registrant under any of the following provisions (see General Instruction A.2.
below):
| ¨ |
Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| ¨ |
Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| ¨ |
Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| ¨ |
Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
| Title of Each Class |
|
Trading Symbol |
|
Name
of Exchange on Which Registered |
| Common Stock, par value $0.0001 per share |
|
IPSC |
|
Nasdaq Global Select Market |
Indicate by check mark whether the registrant is an emerging growth
company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange
Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company x
If an emerging growth company, indicate by check mark if the registrant
has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant
to Section 13(a) of the Exchange Act.
| Item 2.02 |
Results of Operations and Financial Condition |
On August 9, 2023, Century Therapeutics, Inc. (the “Company”)
issued a press release announcing its financial results for the quarter ended June 30, 2023. A copy of the press release is furnished
as Exhibit 99.1 to this Current Report on Form 8-K and is incorporated herein by reference.
The information contained in this Item 2.02 (including Exhibit 99.1)
is being furnished and shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as
amended (the “Exchange Act”), or otherwise subject to the liabilities of that section and shall not be deemed to be
incorporated by reference in any filing under the Securities Act of 1933, as amended (the “Securities Act”), or the
Exchange Act, except as shall be expressly set forth by specific reference in such filing.
| Item 7.01 |
Regulation FD Disclosure |
On August 9, 2023, the Company updated information reflected in a slide presentation, which is attached
as Exhibit 99.2 to this Current Report on Form 8-K and is incorporated herein by reference. Representatives of the Company will use the
updated presentation in various meetings with investors from time to time.
The information contained in this Item 7.01 (including Exhibit
99.2) is being furnished and shall not be deemed "filed" for purposes of Section 18 of the Exchange Act, or otherwise subject to the liabilities
of that section and shall not be deemed to be incorporated by reference in any filing under the Securities Act or the Exchange Act, except
as shall be expressly set forth by specific reference in such filing.
| Item 9.01 |
Financial Statements and Exhibits |
(d) Exhibits
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934,
the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| |
CENTURY THERAPEUTICS, INC. |
| |
|
|
| |
By: |
/s/ Gregory Russotti, Ph.D. |
| |
Name: |
Gregory Russotti, Ph.D. |
| |
Title: |
Interim President and Chief Executive Officer |
Date: August 9, 2022
Exhibit 99.1

Century Therapeutics Reports Second Quarter
2023 Financial Results and Provides Business Updates
– Company remains on track to report initial
data from Schedule A of the ongoing Phase 1 ELiPSE-1 trial of CNTY-101 in relapsed/refractory CD19 positive B-cell lymphomas by year end
–
– Ended second quarter 2023 with cash,
cash equivalents, and investments of $301.0 million; Cash runway expected into 2026 –
PHILADELPHIA, August 9, 2023 -- Century Therapeutics, Inc.
(NASDAQ: IPSC), an innovative clinical-stage biotechnology company developing induced pluripotent stem cell (iPSC)-derived cell therapies
in immuno-oncology, today reported financial results and business highlights for the second quarter ended June 30, 2023.
“Here at Century we have continued to focus on the further advancement
of our pipeline, specifically our ELiPSE-1 Phase 1 trial evaluating CNTY-101 in relapsed or refractory CD19 positive B-cell lymphomas,
for which we expect to report initial data from Schedule A by year end,” said Greg Russotti, Ph.D., Interim Chief Executive
Officer, Century Therapeutics. “We remain confident in our differentiated scientific approach and are looking forward to spending
the second half of this year working towards solidifying our position as an innovative leader in the cell therapy space.”
Business Highlights & Upcoming Milestones
| ● | The first-in-human Phase 1 ELiPSE-1 trial evaluating CNTY-101 in relapsed or refractory CD19 positive B-cell lymphomas is ongoing.
The Company remains on track to report preliminary data from Schedule A of the trial, including pharmacokinetics, pharmacodynamics, and
safety, by year end. |
| ● | At the American Society
for Clinical Oncology (ASCO) Annual Meeting in June 2023, the Company presented a Trials
in Progress poster related to its Phase 1 ELiPSE-1 trial. A copy of the poster, titled, “The
ELiPSE-1 Study: A Phase 1 Multicenter Open-Label Study of CNTY-101 in Subjects with Relapsed
or Refractory CD19-Positive B Cell Malignancies”, is available on the Posters section
of Century’s website at https://www.centurytx.com/science/. |
Second Quarter 2023 Financial Results
| ● | Cash Position: Cash, cash equivalents, and marketable securities were $301.0 million as of June 30, 2023, as compared
to $367.4 million as of December 31, 2022. Net cash used in operations was $48.5 million for the six months ended June 30, 2023,
compared to net cash provided by operations of $61.2 million for the six months ended June 30, 2022 (which includes deferred revenue
from the Bristol Myers Squibb (BMS) collaboration of $120.7 million). |

| ● | Collaboration Revenue: Collaboration revenue generated through the Company’s collaboration, option and license agreement
with BMS was $0.1 million for the three months ended June 30, 2023, compared to $1.4 million for the same period in 2022. |
| ● | Research and Development (R&D) expenses: R&D expenses were $22.7 million for the three months ended June 30, 2023,
compared to $24.5 million for the same period in 2022. The decrease in R&D expenses was primarily due to the reduction in force in
January of 2023. |
| ● | General and Administrative (G&A) expenses: G&A expenses were $8.2 million for the three months ended June 30,
2023, compared to $8.3 million for the same period in 2022. The decrease in G&A expenses was primarily due to a reduction in headcount. |
| ● | Impairment of long lived assets: A one-time impairment charge of $4.2 million was recorded in connection with the strategic
decision to consolidate two of the Company’s existing leased facilities in Philadelphia. |
| ● | Net loss: Net loss was $33.3 million for the three months ended June 30, 2023, compared to $31.0 million for the three
months ended June 30, 2022. |
Financial Guidance
| ● | The Company expects full year generally accepted accounting principles (GAAP) operating expenses to be between $135 million and $145
million, including non-cash stock-based compensation expense of $12 million to $17 million. |
| ● | The Company estimates its cash, cash equivalents, and investments will support operations into 2026. |
About Century Therapeutics
Century Therapeutics (NASDAQ: IPSC) is harnessing
the power of adult stem cells to develop curative cell therapy products for cancer that we believe will allow us to overcome the limitations
of first-generation cell therapies. Our genetically engineered, iPSC-derived iNK and iT cell product candidates are designed to specifically
target hematologic and solid tumor cancers. We are leveraging our expertise in cellular reprogramming, genetic engineering, and manufacturing
to develop therapies with the potential to overcome many of the challenges inherent to cell therapy and provide a significant advantage
over existing cell therapy technologies. We believe our commitment to developing off-the-shelf cell therapies will expand patient access
and provide an unparalleled opportunity to advance the course of cancer care. For more information on Century Therapeutics please visit
www.centurytx.com.

Century Therapeutics Forward-Looking Statement
This press release contains forward-looking statements
within the meaning of, and made pursuant to the safe harbor provisions of, The Private Securities Litigation Reform Act of 1995. All statements
contained in this press release, other than statements of historical facts or statements that relate to present facts or current conditions,
including but not limited to, statements regarding our clinical development plans and timelines, are forward-looking statements. These
statements involve known and unknown risks, uncertainties and other important factors that may cause our actual results, performance,
or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking
statements. In some cases, you can identify forward-looking statements by terms such as “may,” “might,” “will,”
“should,” “expect,” “plan,” “aim,” “seek,” “anticipate,” “could,”
“intend,” “target,” “project,” “contemplate,” “believe,” “estimate,”
“predict,” “forecast,” “potential” or “continue” or the negative of these terms or other
similar expressions. The forward-looking statements in this press release are only predictions. We have based these forward-looking statements
largely on our current expectations and projections about future events and financial trends that we believe may affect our business,
financial condition, and results of operations. These forward-looking statements speak only as of the date of this press release and are
subject to a number of risks, uncertainties and assumptions, some of which cannot be predicted or quantified and some of which are beyond
our control, including, among others: our ability to successfully advance our current and future product candidates through development
activities, preclinical studies, and clinical trials; our dependence on the success of our lead product candidate, CNTY-101,our ability
to obtain FDA acceptance for our future IND submissions and commence clinical trials on expected timelines, or at all; our reliance on
the maintenance of certain key collaborative relationships for the manufacturing and development of our product candidates; the timing,
scope and likelihood of regulatory filings and approvals, including final regulatory approval of our product candidates; the impact of
the COVID-19 pandemic, geopolitical issues, banking instability and inflation on our business and operations, supply chain and labor force;
the performance of third parties in connection with the development of our product candidates, including third parties conducting our
clinical trials as well as third-party suppliers and manufacturers; our ability to successfully commercialize our product candidates and
develop sales and marketing capabilities, if our product candidates are approved; our ability to recruit and maintain key members of management
and our ability to maintain and successfully enforce adequate intellectual property protection. These and other risks and uncertainties
are described more fully in the “Risk Factors” section of our most recent filings with the Securities and Exchange Commission
and available at www.sec.gov. You should not rely on these forward-looking statements as predictions of future events. The events and
circumstances reflected in our forward-looking statements may not be achieved or occur, and actual results could differ materially from
those projected in the forward-looking statements. Moreover, we operate in a dynamic industry and economy. New risk factors and uncertainties
may emerge from time to time, and it is not possible for management to predict all risk factors and uncertainties that we may face. Except
as required by applicable law, we do not plan to publicly update or revise any forward-looking statements contained herein, whether as
a result of any new information, future events, changed circumstances or otherwise.
For More Information:
Investors and Media: Melissa Forst/Maghan Meyers – century@argotpartners.com

Century Therapeutics, Inc
Condensed Balance Sheets
(unaudited, in thousands)
| | |
June 30, | | |
December 31, | |
| |
2023 | | |
2022 | |
| Assets | |
|
| | |
|
| |
| Current Assets: | |
| | | |
| | |
| Cash and cash equivalents | |
$ | 64,439 | | |
$ | 84,265 | |
| Short-term investments | |
| 91,958 | | |
| 231,233 | |
| Prepaid expenses and other current assets | |
| 4,883 | | |
| 4,223 | |
| Total current assets | |
| 161,280 | | |
| 319,721 | |
| Property and equipment, net | |
| 83,771 | | |
| 82,785 | |
| Operating lease right-of-use assets, net | |
| 24,993 | | |
| 28,945 | |
| Long-term investments | |
| 144,581 | | |
| 51,854 | |
| Other long-term assets | |
| 2,534 | | |
| 3,239 | |
| Total assets | |
$ | 417,159 | | |
$ | 486,544 | |
| | |
| | | |
| | |
| Liabilities, convertible preferred stock, and stockholders' equity | |
| | | |
| | |
| Current liabilities: | |
| | | |
| | |
| Accounts payable | |
$ | 3,625 | | |
$ | 5,454 | |
| Accrued expenses and other liabilities | |
| 8,600 | | |
| 10,707 | |
| Long-term debt, current | |
| - | | |
| 6,502 | |
| Deferred revenue, current | |
| 6,936 | | |
| 7,154 | |
| Total current liabilities | |
| 19,161 | | |
| 29,817 | |
| Operating lease liability, noncurrent | |
| 40,833 | | |
| 38,698 | |
| Long-term debt, net | |
| - | | |
| 3,739 | |
| Other long-term liabilities | |
| 361 | | |
| 718 | |
| Deferred revenue | |
| 109,233 | | |
| 110,834 | |
| Total liabilities | |
| 169,588 | | |
| 183,806 | |
| Stockholders' equity | |
| | | |
| | |
| Common stock | |
| 6 | | |
| 6 | |
| Additional paid-in capital | |
| 832,425 | | |
| 824,292 | |
| Accumulated deficit | |
| (583,653 | ) | |
| (519,098 | ) |
| Accumulated other comprehensive loss | |
| (1,207 | ) | |
| (2,462 | ) |
| Total stockholders' equity | |
| 247,571 | | |
| 302,738 | |
| Total liabilities and stockholders' equity | |
$ | 417,159 | | |
$ | 486,544 | |

Century Therapeutics, Inc
Condensed consolidated statements of operations
(unaudited, in thousands, except share and per share amounts)
| | |
Three months Ended | | |
Six months Ended | |
| | |
June 30, | | |
June 30, | | |
June 30, | | |
June 30, | |
| | |
2023 | | |
2022 | | |
2023 | | |
2022 | |
| Collaboration Revenue | |
$ | 99 | | |
$ | 1,396 | | |
$ | 1,819 | | |
$ | 2,454 | |
| | |
| | | |
| | | |
| | | |
| | |
| Operating Expenses | |
| | | |
| | | |
| | | |
| | |
| Research and development | |
$ | 22,727 | | |
$ | 24,494 | | |
$ | 47,626 | | |
$ | 45,690 | |
| General and administrative | |
| 8,229 | | |
| 8,253 | | |
| 17,131 | | |
| 15,551 | |
| In-process research and development | |
| - | | |
| - | | |
| - | | |
| 10,000 | |
| Impairment on long-lived assets | |
| 4,220 | | |
| - | | |
| 4,220 | | |
| - | |
| Total operating expenses | |
$ | 35,176 | | |
$ | 32,747 | | |
$ | 68,977 | | |
$ | 71,241 | |
| | |
| | | |
| | | |
| | | |
| | |
| Loss from operations | |
| (35,077 | ) | |
| (31,351 | ) | |
| (67,158 | ) | |
| (68,787 | ) |
| | |
| | | |
| | | |
| | | |
| | |
| Interest expense | |
| (136 | ) | |
| (330 | ) | |
| (540 | ) | |
| (644 | ) |
| Interest income | |
| 3,058 | | |
| 706 | | |
| 5,681 | | |
| 960 | |
| Other income, net | |
| (186 | ) | |
| 5 | | |
| (380 | ) | |
| 4 | |
| Loss before provision for income taxes | |
$ | (32,341 | ) | |
$ | (30,970 | ) | |
$ | (62,397 | ) | |
$ | (68,467 | ) |
| Provision for income taxes | |
| (950 | ) | |
| (18 | ) | |
| (2,158 | ) | |
| (34 | ) |
| Net Loss | |
$ | (33,291 | ) | |
$ | (30,988 | ) | |
$ | (64,555 | ) | |
$ | (68,501 | ) |
| | |
| | | |
| | | |
| | | |
| | |
| Unrealized loss on investments | |
| 59 | | |
| (780 | ) | |
| 1,255 | | |
| (2,766 | ) |
| Foreign currency translation adjustment | |
| 9 | | |
| (12 | ) | |
| - | | |
| (18 | ) |
| Comprehensive loss | |
| (33,223 | ) | |
| (31,780 | ) | |
| (63,300 | ) | |
| (71,285 | ) |
| | |
| | | |
| | | |
| | | |
| | |
| Net loss per common share - Basic and Diluted | |
| (0.56 | ) | |
| (0.54 | ) | |
| (1.10 | ) | |
| (1.19 | ) |
| | |
| | | |
| | | |
| | | |
| | |
| Weighted average common shares outstanding | |
| 59,251,363 | | |
| 57,685,006 | | |
| 58,904,726 | | |
| 57,370,022 | |
Exhibit 99.2
| Corporate Overview
Aug 2023 |

| 2
Forward-looking statements
This presentation contains forward-looking statements within the meaning
of, and made pursuant to the safe harbour provisions of, The Private
Securities Litigation Reform Act of 1995. All statements contained in this
document, other than statements of historical facts or statements that
relate to present facts or current conditions, including but not limited to,
statements regarding possible or assumed future results of operations,
business strategies, research and development plans, regulatory activities,
market opportunity, competitive position and potential growth
opportunities are forward-looking statements. These statements involve
known and unknown risks, uncertainties and other important factors that
may cause our actual results, performance or achievements to be materially
different from any future results, performance or achievements expressed
or implied by the forward-looking statements. In some cases, you can
identify forward-looking statements by terms such as “may,” “might,” “will,”
“should,” “expect,” “plan,” “aim,” “seek,” “anticipate,” “could,” “intend,”
“target,” “project,” “contemplate,” “believe,” “estimate,” “predict,” “forecast,”
“potential” or “continue” or the negative of these terms or other similar
expressions. The forward-looking statements in this presentation are only
predictions. We have based these forward-looking statements largely on
our current expectations and projections about future events and financial
trends that we believe may affect our business, financial condition and
results of operations. These forward-looking statements speak only as of the
date of this presentation and are subject to a number of risks, uncertainties
and assumptions, some of which cannot be predicted or quantified and
some of which are beyond our control, including, among others: our ability
to successfully advance our current and future product candidates through
development activities, preclinical studies, and clinical trials; our reliance
on the maintenance on certain key collaborative relationships for the
manufacturing and development of our product candidates; the timing,
scope and likelihood of regulatory filings and approvals, including final
regulatory approval of our product candidates; the impact of the COVID-19
pandemic, geopolitical issues and inflation on our business and
operations, supply chain and labor force; the performance of third parties
in connection with the development of our product candidates, including
third parties conducting our future clinical trials as well as third-party
suppliers and manufacturers; our ability to successfully commercialize our
product candidates and develop sales and marketing capabilities, if our
product candidates are approved; and our ability to maintain and
successfully enforce adequate intellectual property protection. These and
other risks and uncertainties are described more fully in the “Risk Factors”
section of our most recent filings with the Securities and Exchange
Commission and available at www.sec.gov. You should not rely on these
forward-looking statements as predictions of future events. The events
and circumstances reflected in our forward-looking statements may not
be achieved or occur, and actual results could differ materially from those
projected in the forward-looking statements. Moreover, we operate in a
dynamic industry and economy. New risk factors and uncertainties may
emerge from time to time, and it is not possible for management to
predict all risk factors and uncertainties that we may face. Except as
required by applicable law, we do not plan to publicly update or revise any
forward-looking statements contained herein, whether as a result of any
new information, future events, changed circumstances or otherwise. |

| 3
Investment Thesis
Next generation platforms for iNK and gamma delta iT candidates
Foundational investments in iPSC technology, genetic editing, and manufacturing
Experienced team in R&D, immuno-oncology, manufacturing and
commercialization
Exemplified by FDA clearance of Century’s first IND for CNTY-101 & trial execution
Well capitalized with cash runway into 2026
$301.0M in cash, cash equivalents and investments at the end of 2Q23; operational
efficiencies designed to enable delivery on key milestones, clinical data |

| iPSC Platform |
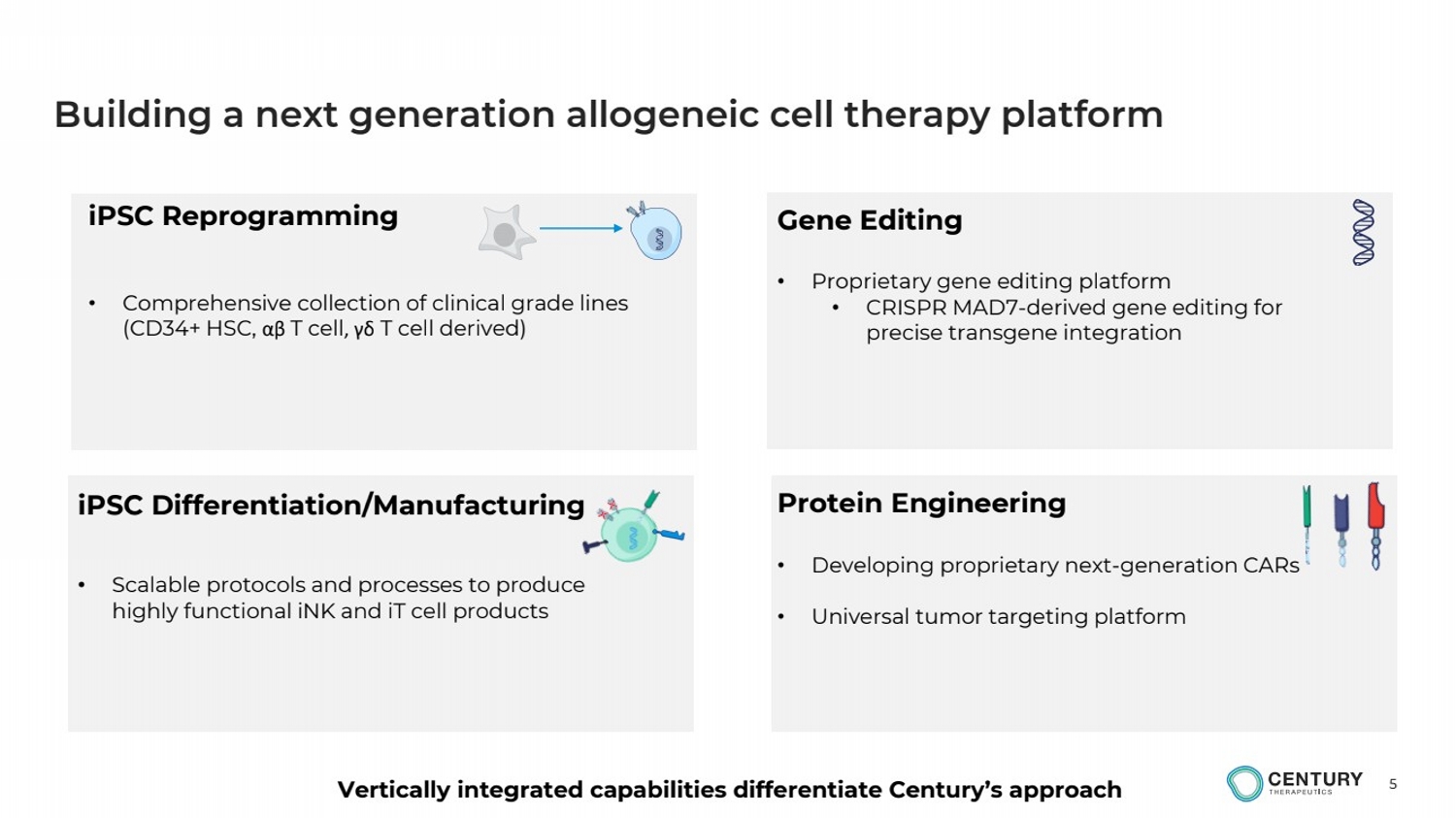
| 5
Building a next generation allogeneic cell therapy platform
Gene Editing
• Proprietary gene editing platform
• CRISPR MAD7-derived gene editing for
precise transgene integration
Protein Engineering
• Developing proprietary next-generation CARs
• Universal tumor targeting platform
iPSC Differentiation/Manufacturing
• Scalable protocols and processes to produce
highly functional iNK and iT cell products
iPSC Reprogramming
• Comprehensive collection of clinical grade lines
(CD34+ HSC, αβ T cell, γδ T cell derived)
Vertically integrated capabilities differentiate Century’s approach |

| 6
Foundational investments in iPSC know-how
and manufacturing
Established in-house manufacturing
accelerates learnings and enables faster
product iteration
• 53,000 ft2
facility
• Designed to produce multiple immune cell types
• Two sites provides optionality and maximizes
flexibility
iPSC license and collaboration agreement
established in 2018
• Access to clinical grade iPSC lines
• Exclusive IP and know-how to generate immune
effector cells using feeder-free methods (NK, T,
Mac, DC)
• FCDI GMP manufacturing capacity for Century’s
product candidates
• Leveraging two decades of research & investment
at University of Wisconsin and FCDI |
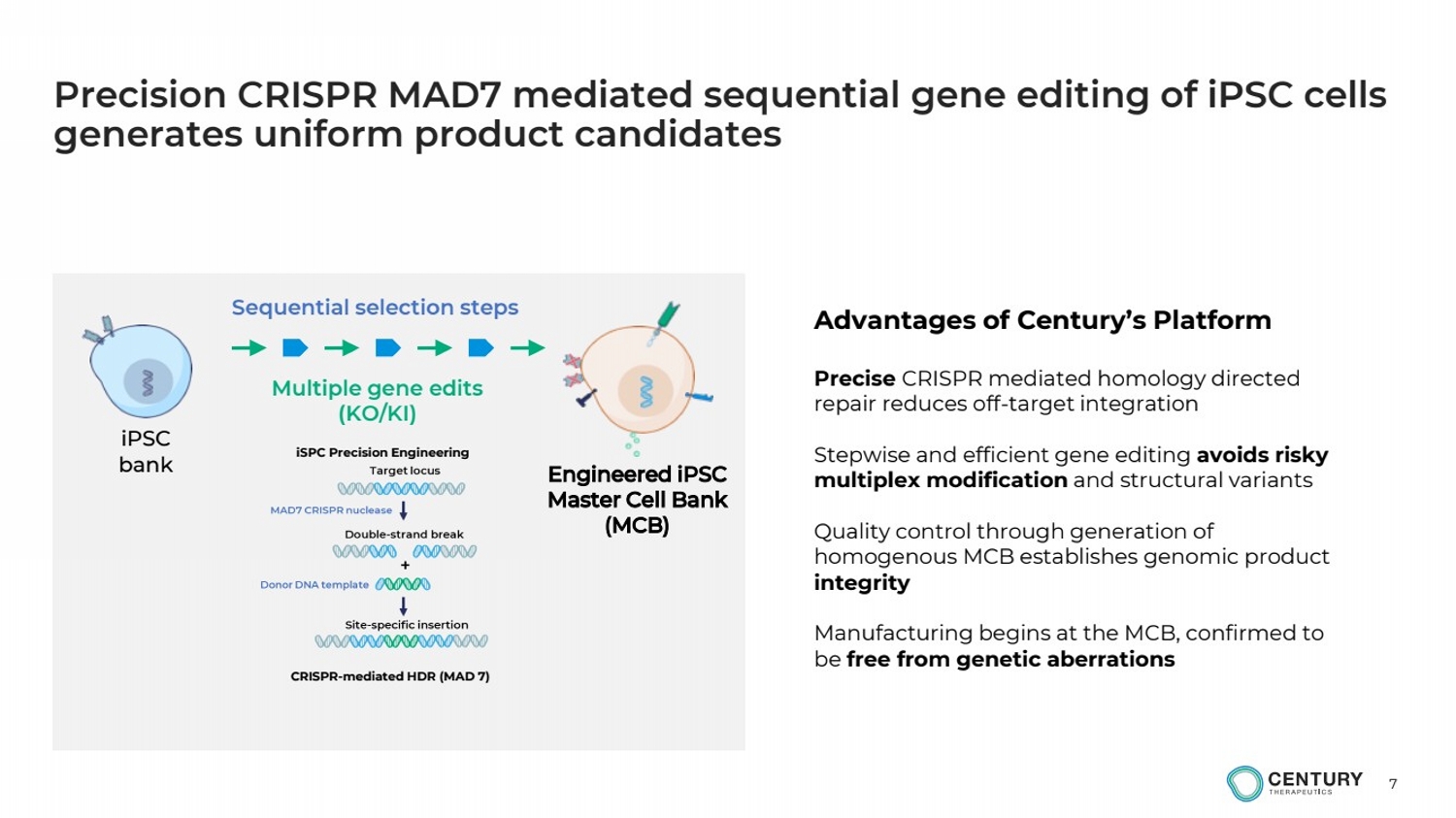
| 7
Multiple gene edits
(KO/KI)
iPSC
bank
Precision CRISPR MAD7 mediated sequential gene editing of iPSC cells
generates uniform product candidates
Engineered iPSC
Master Cell Bank
(MCB)
Advantages of Century’s Platform
Precise CRISPR mediated homology directed
repair reduces off-target integration
Stepwise and efficient gene editing avoids risky
multiplex modification and structural variants
Quality control through generation of
homogenous MCB establishes genomic product
integrity
Manufacturing begins at the MCB, confirmed to
be free from genetic aberrations
Sequential selection steps
iSPC Precision Engineering
CRISPR-mediated HDR (MAD 7) |
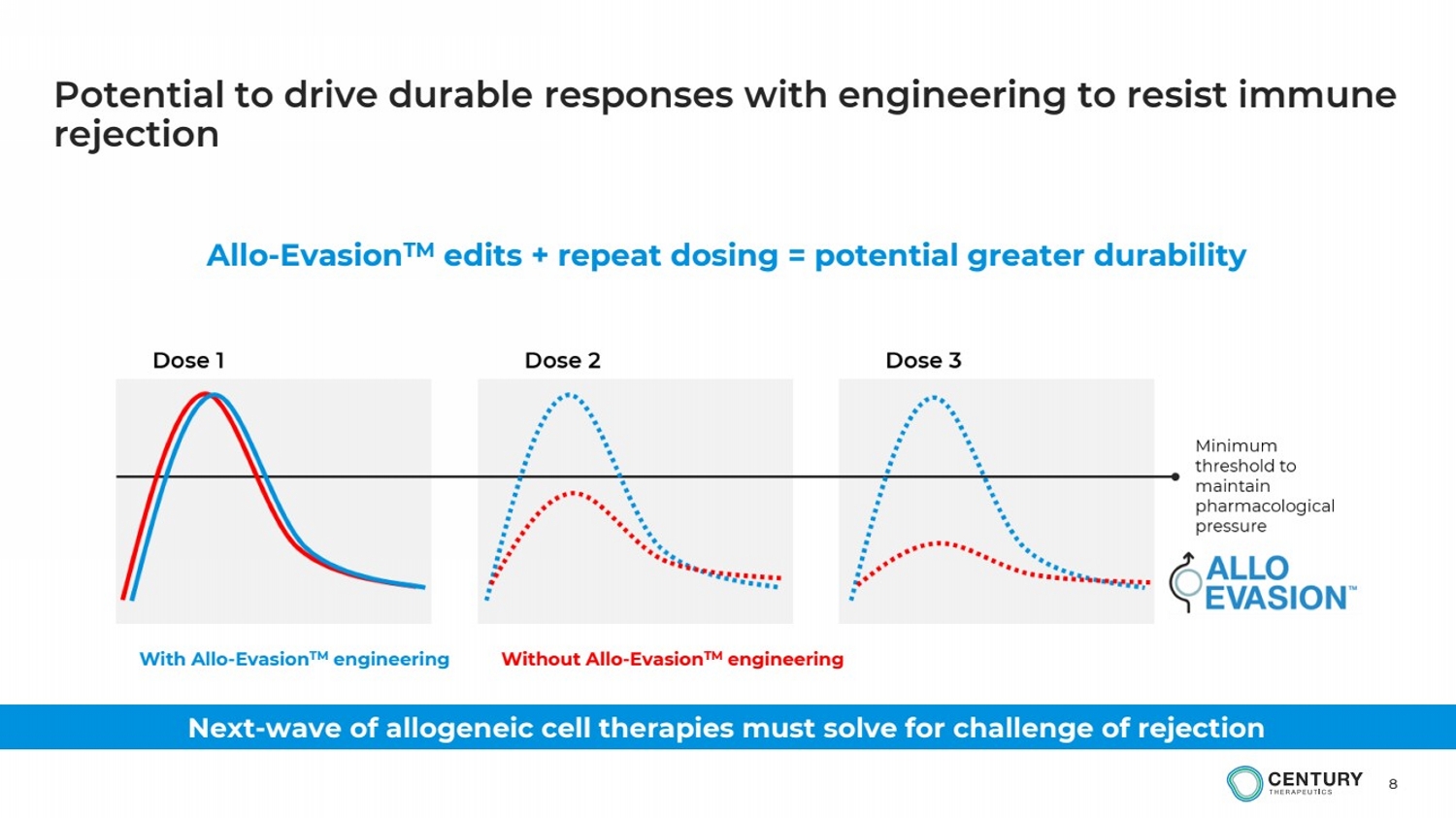
| 8
Potential to drive durable responses with engineering to resist immune
rejection
Allo-EvasionTM edits + repeat dosing = potential greater durability
Next-wave of allogeneic cell therapies must solve for challenge of rejection
With Allo-EvasionTM engineering Without Allo-EvasionTM engineering |
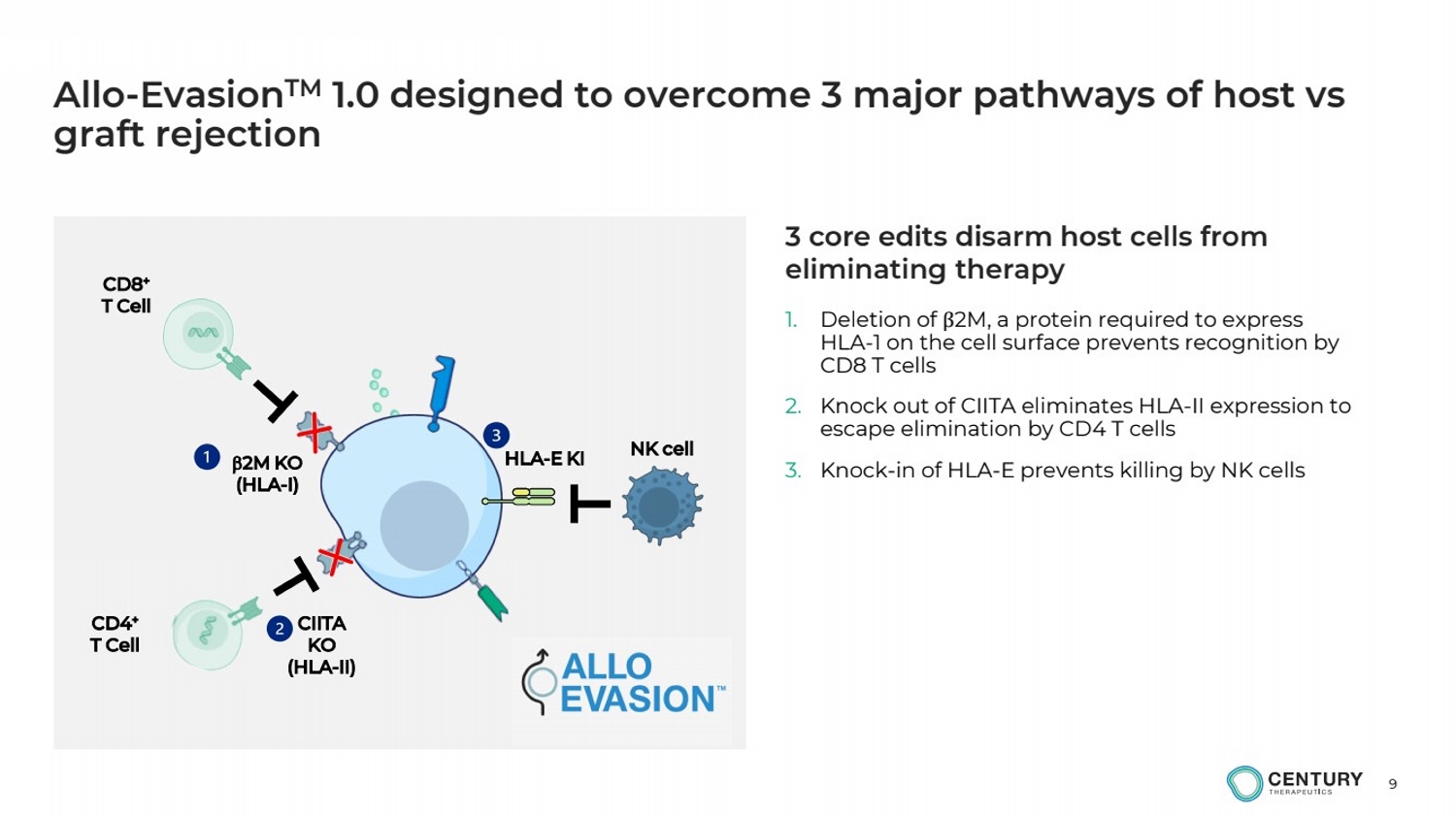
| 9
Allo-EvasionTM 1.0 designed to overcome 3 major pathways of host vs
graft rejection
1. Deletion of β2M, a protein required to express
HLA-1 on the cell surface prevents recognition by
CD8 T cells
2. Knock out of CIITA eliminates HLA-II expression to
escape elimination by CD4 T cells
3. Knock-in of HLA-E prevents killing by NK cells b2M KO
(HLA-I)
HLA-E KI
CIITA
KO
(HLA-II)
CD8+
T Cell
CD4+
T Cell
NK cell
3 core edits disarm host cells from
eliminating therapy |
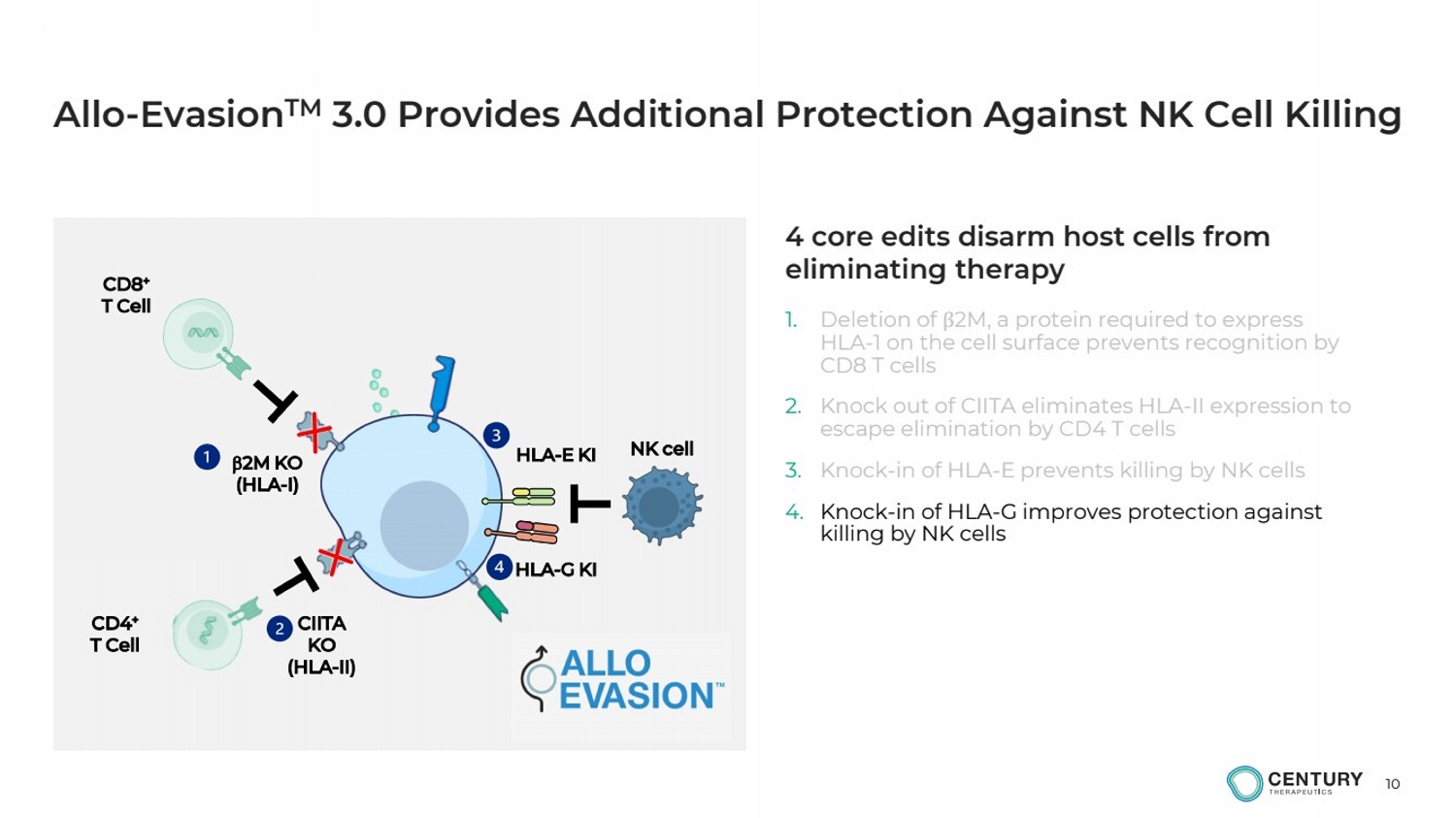
| 10
Allo-EvasionTM 3.0 Provides Additional Protection Against NK Cell Killing
1. Deletion of β2M, a protein required to express
HLA-1 on the cell surface prevents recognition by
CD8 T cells
2. Knock out of CIITA eliminates HLA-II expression to
escape elimination by CD4 T cells
3. Knock-in of HLA-E prevents killing by NK cells
4. Knock-in of HLA-G improves protection against
killing by NK cells
b2M KO
(HLA-I)
HLA-E KI
CIITA
KO
(HLA-II)
CD8+
T Cell
CD4+
T Cell
NK cell
4 core edits disarm host cells from
eliminating therapy
HLA-G KI |
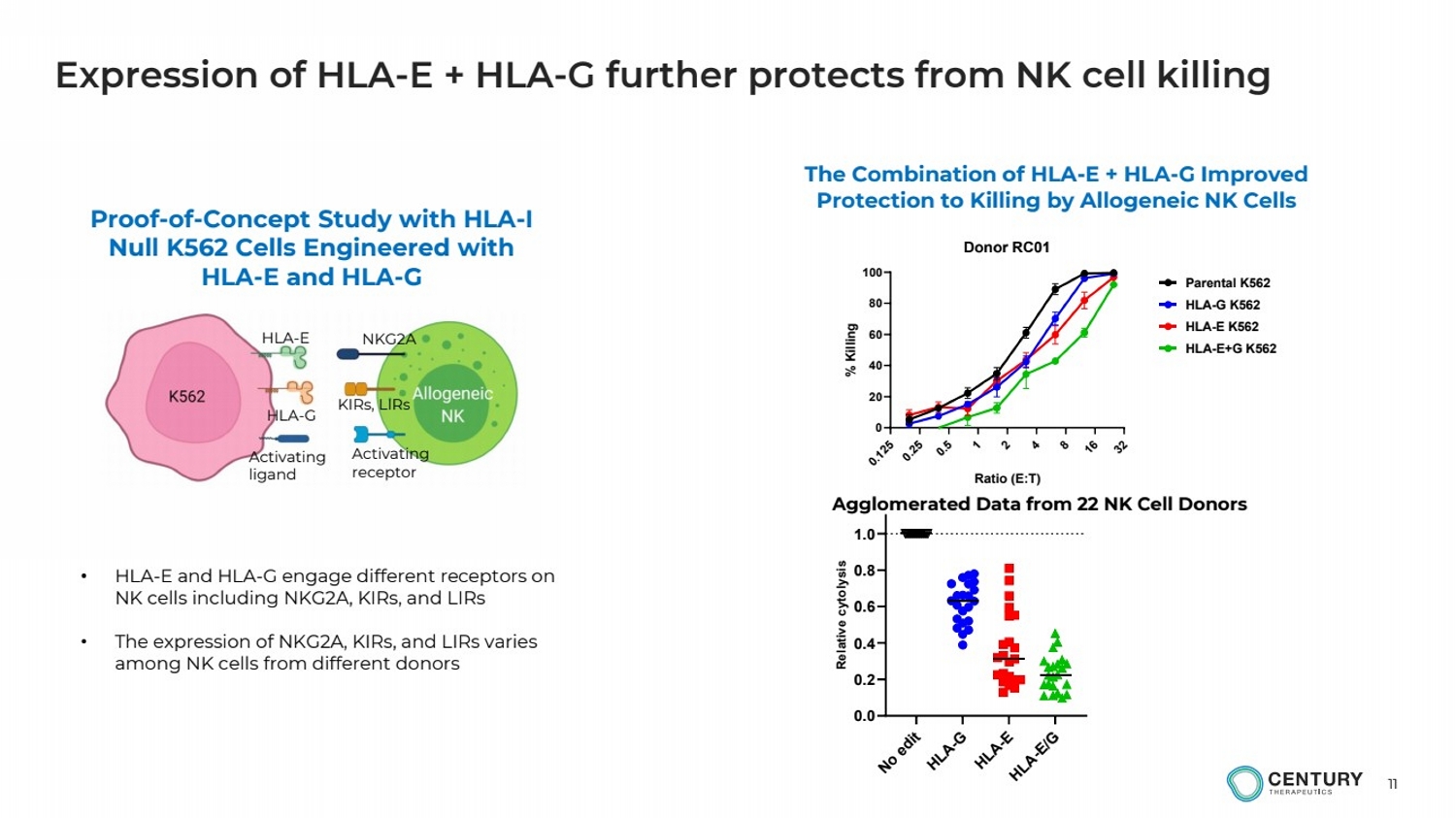
| 11
Expression of HLA-E + HLA-G further protects from NK cell killing
HLA-E
HLA-G
NKG2A
KIRs, LIRs
Activating
ligand
Activating
receptor
Proof-of-Concept Study with HLA-I
Null K562 Cells Engineered with
HLA-E and HLA-G
0.125
0.25
0.5
1
2
4
8
16
32
0
20
40
60
80
100 Donor RC01
Ratio (E:T)
% Killing
Parental K562 HLA-G K562 HLA-E K562 HLA-E+G K562
The Combination of HLA-E + HLA-G Improved
Protection to Killing by Allogeneic NK Cells
• HLA-E and HLA-G engage different receptors on
NK cells including NKG2A, KIRs, and LIRs
• The expression of NKG2A, KIRs, and LIRs varies
among NK cells from different donors
Agglomerated Data from 22 NK Cell Donors
No edit HLA-G
HLA-E HLA-E/G
0.0
0.2
0.4
0.6
0.8
1.0
Relative cytolysis |

| Pipeline |
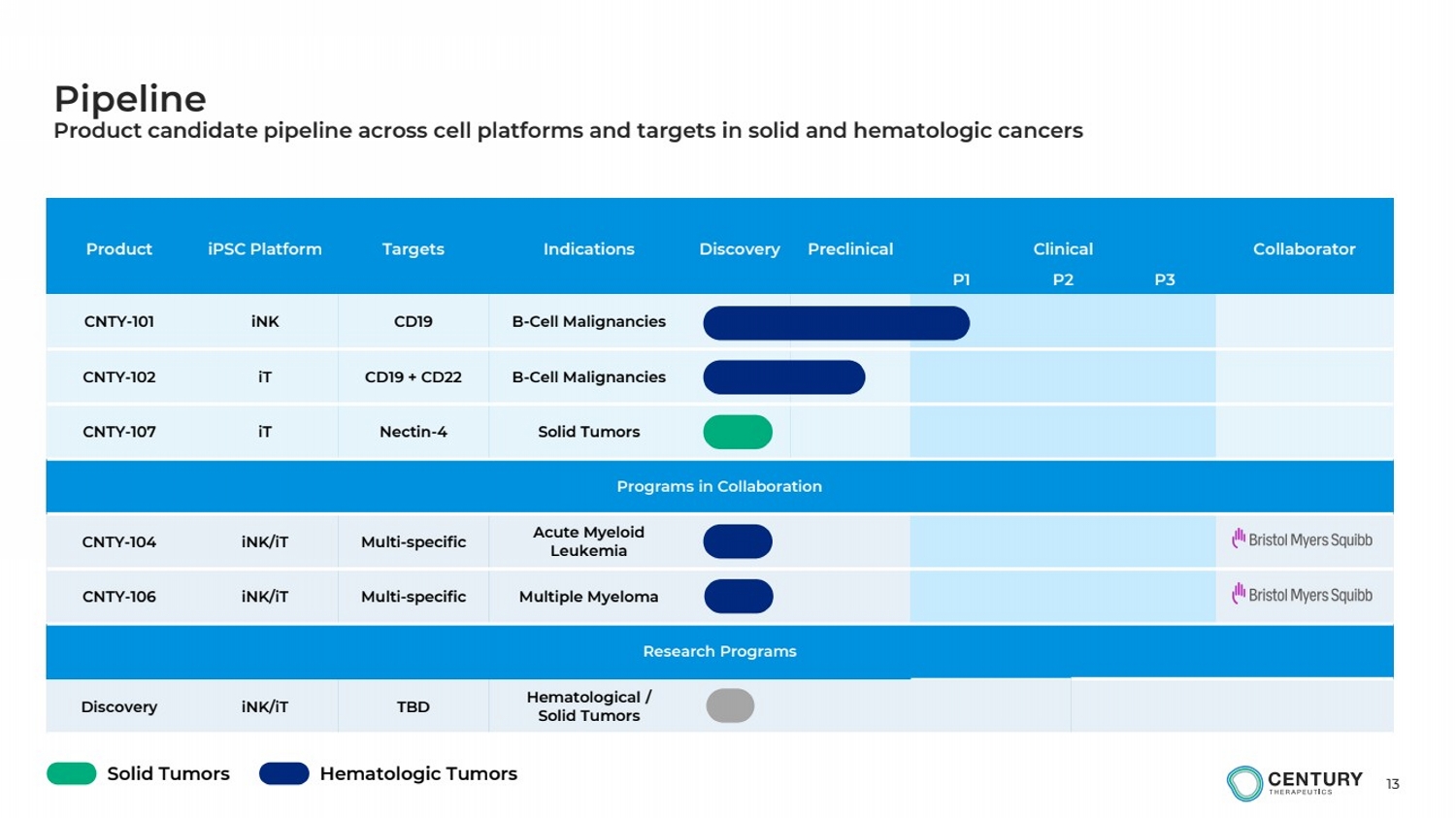
| 13
Product iPSC Platform Targets Indications Discovery Preclinical Clinical Collaborator
P1 P2 P3
CNTY-101 iNK CD19 B-Cell Malignancies
CNTY-102 iT CD19 + CD22 B-Cell Malignancies
CNTY-107 iT Nectin-4 Solid Tumors
Programs in Collaboration
CNTY-104 iNK/iT Multi-specific Acute Myeloid
Leukemia
CNTY-106 iNK/iT Multi-specific Multiple Myeloma
Research Programs
Discovery iNK/iT TBD Hematological /
Solid Tumors
Pipeline
Product candidate pipeline across cell platforms and targets in solid and hematologic cancers
Solid Tumors Hematologic Tumors |
| 14
Promise of allogeneic cell therapies in lymphoma
Large unmet need remains despite
progress with autologous cell
therapies
• ~25% of eligible patients receive
CAR-T therapy1
• ~35% of patients achieve long-term remission even in earlier
lines of therapy1
Off-the-shelf modalities
approaching bar set by autologous
but falling short on durability
• Rejection limits potential of
durable responses for first wave of
allogeneic cell products
• Bispecifics lack curative potential
of cell therapy
Goal to deliver more durable
response rates vs autologous
X
• Century candidates designed to
realize benefit of repeat dosing
enabled by Allo-EvasionTM
• Shift from “one and done” to finite
repeat dosing to increase
pharmacological pressure
1. Targeted Oncology, Many Challenges, Opportunities for CAR T-Cell Therapies in Lymphoma, Sept 2022 |
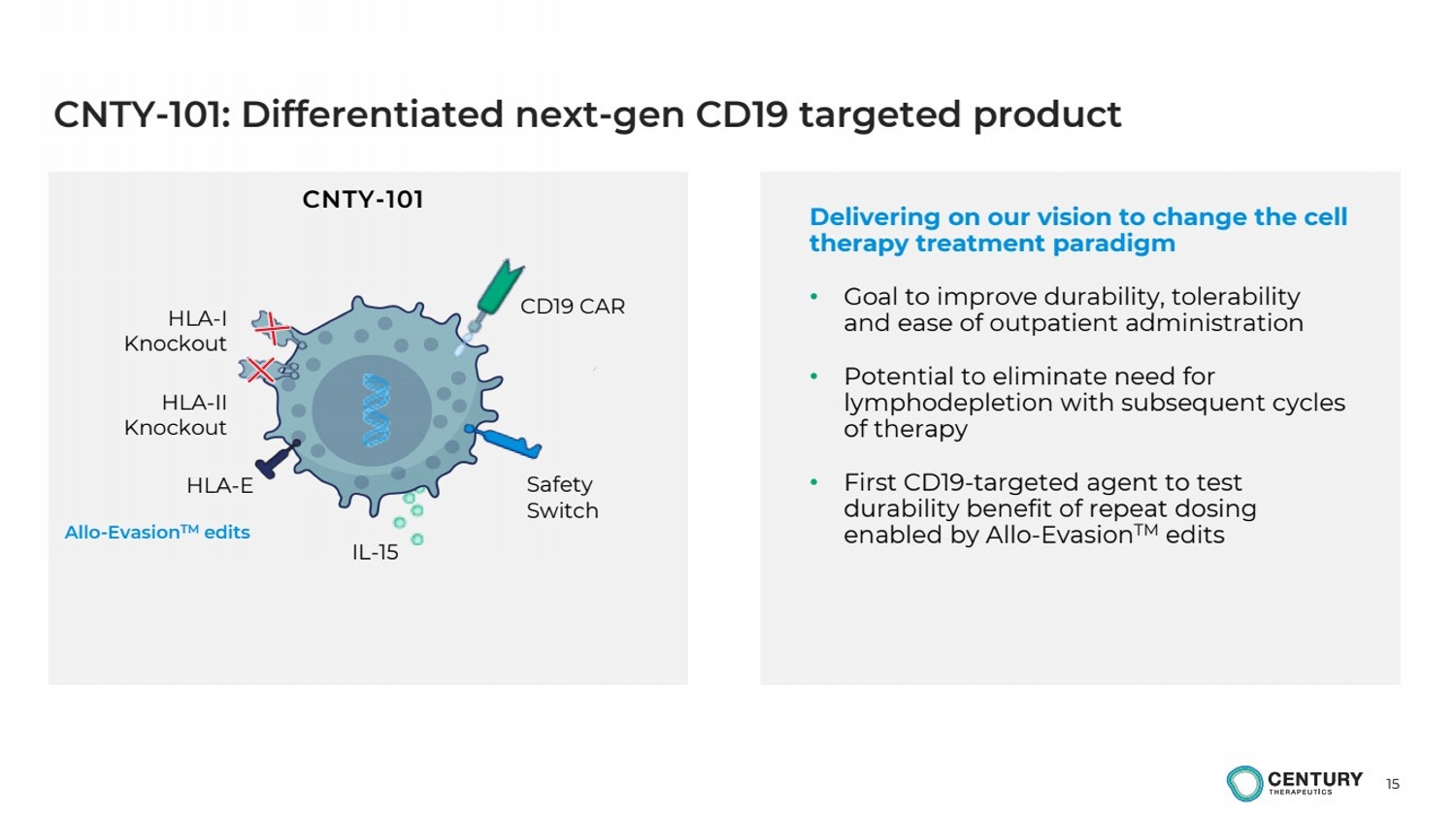
| 15
Delivering on our vision to change the cell
therapy treatment paradigm
• Goal to improve durability, tolerability
and ease of outpatient administration
• Potential to eliminate need for
lymphodepletion with subsequent cycles
of therapy
• First CD19-targeted agent to test
durability benefit of repeat dosing
enabled by Allo-EvasionTM edits
CNTY-101: Differentiated next-gen CD19 targeted product
CNTY-101
Allo-EvasionTM edits
HLA-I
Knockout
IL-15
HLA-II
Knockout
CD19 CAR
HLA-E Safety
Switch |
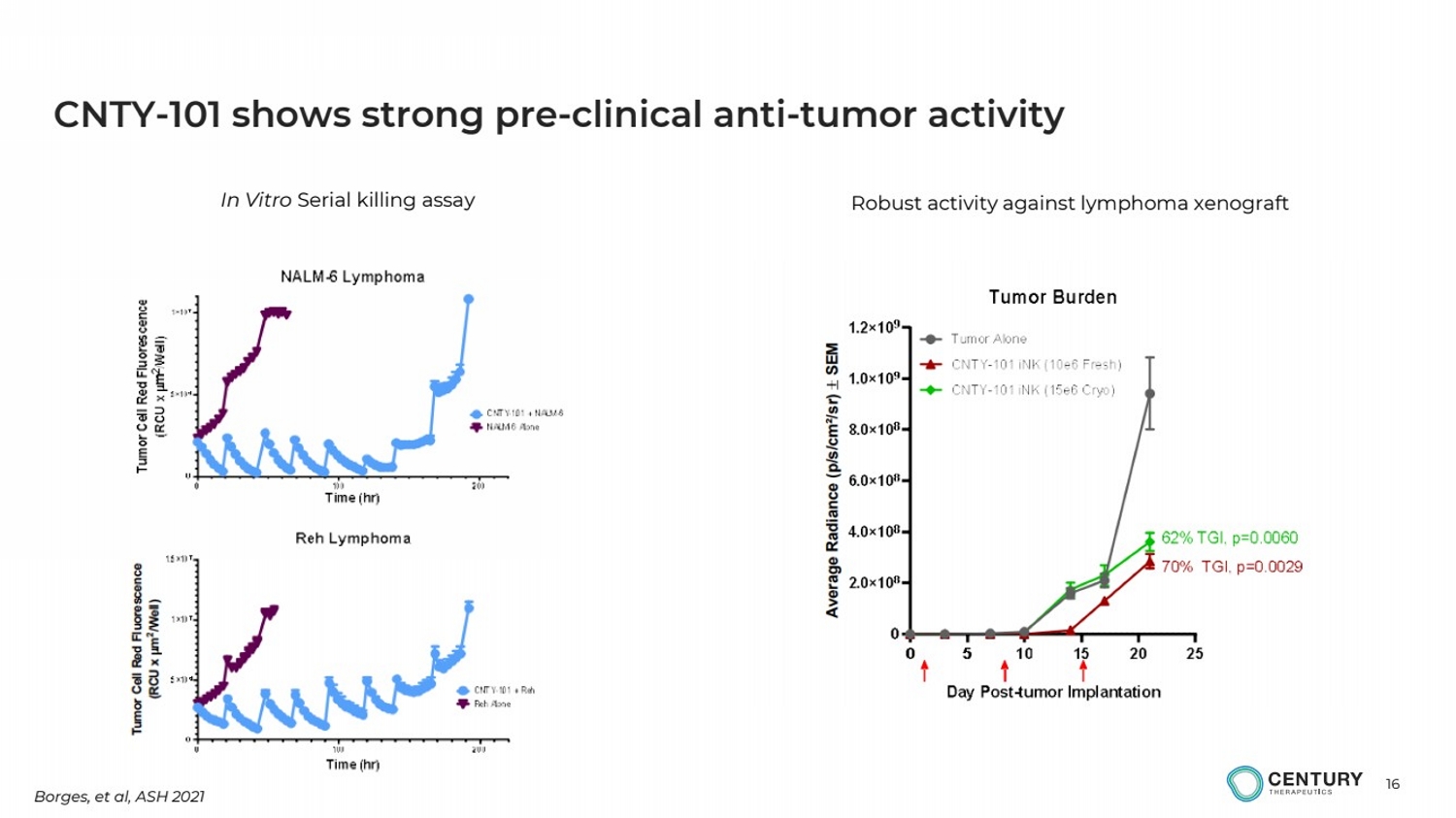
| 16
CNTY-101 shows strong pre-clinical anti-tumor activity
In Vitro Serial killing assay Robust activity against lymphoma xenograft
Borges, et al, ASH 2021 |
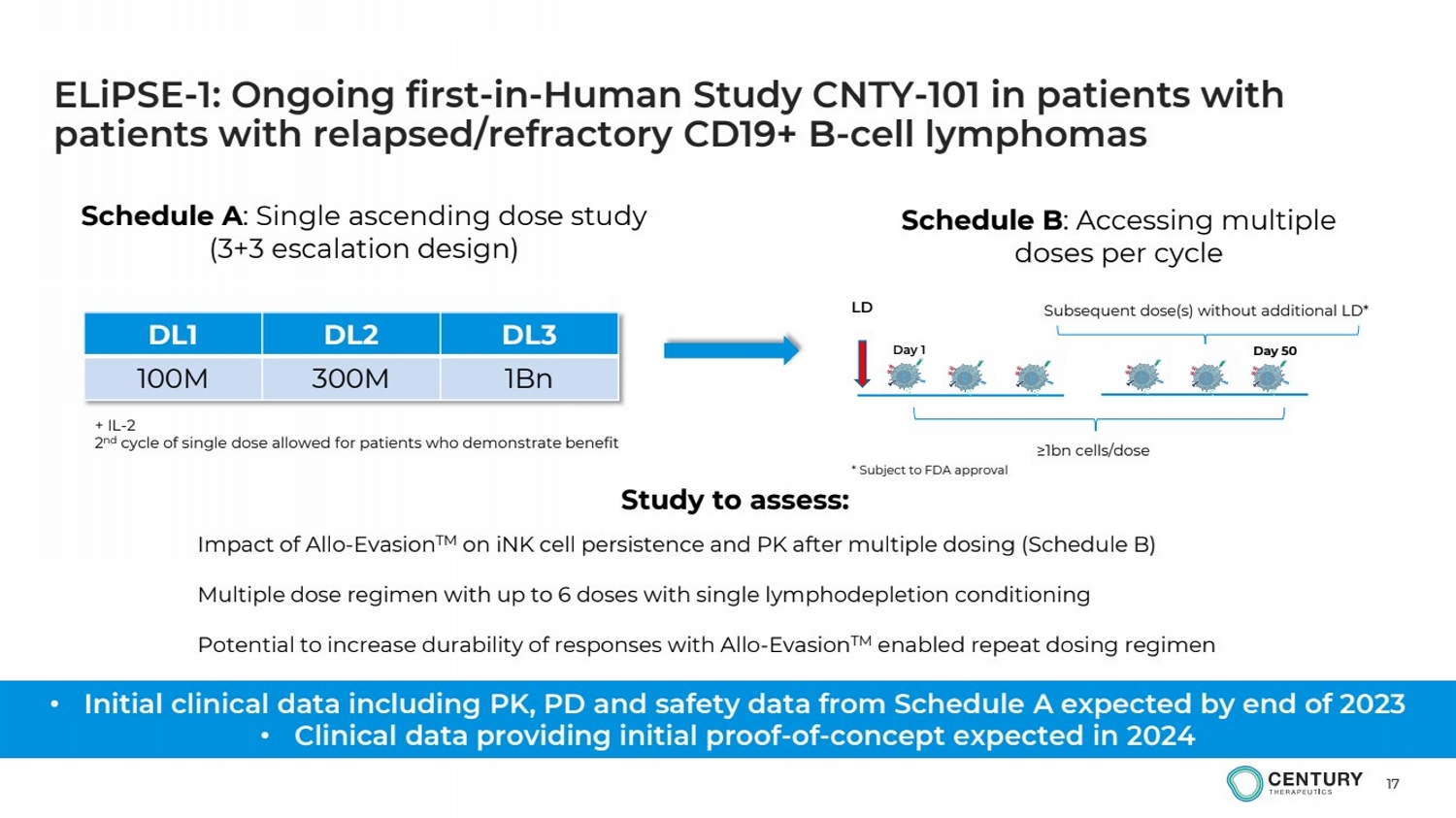
| 17
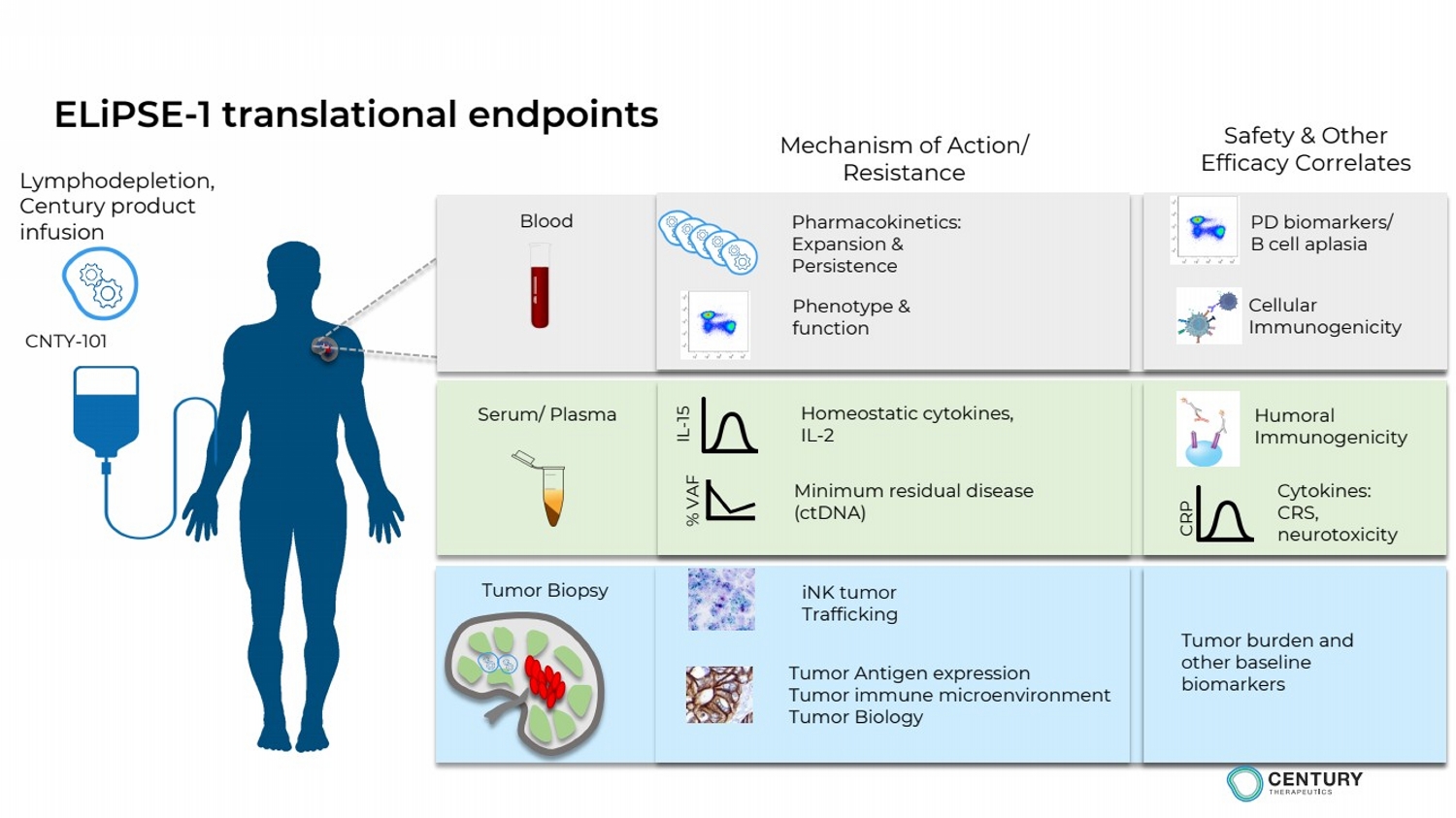
ELiPSE-1: Ongoing first-in-Human Study CNTY-101 in patients with
patients with relapsed/refractory CD19+ B-cell lymphomas
Study to assess:
Impact of Allo-EvasionTM on iNK cell persistence and PK after multiple dosing (Schedule B)
Multiple dose regimen with up to 6 doses with single lymphodepletion conditioning
Potential to increase durability of responses with Allo-EvasionTM enabled repeat dosing regimen
Day 1
LD
Day 50
Subsequent dose(s) without additional LD*
≥1bn cells/dose
* Subject to FDA approval
Schedule A: Single ascending dose study
(3+3 escalation design)
DL1 DL2 DL3
100M 300M 1Bn
Schedule B: Accessing multiple
doses per cycle
+ IL-2
2
nd cycle of single dose allowed for patients who demonstrate benefit
• Initial clinical data including PK, PD and safety data from Schedule A expected by end of 2023
• Clinical data providing initial proof-of-concept expected in 2024 |
| t
8
,
2
0
2
3
1
8
Lymphodepletion,
Century product
infusion
CNTY-101
CRP
IL-15 % VAF
Blood
Serum/ Plasma
Tumor Biopsy
Mechanism of Action/
Resistance
Safety & Other
Efficacy Correlates
Pharmacokinetics:
Expansion &
Persistence
Phenotype &
function
Homeostatic cytokines,
IL-2
Minimum residual disease
(ctDNA)
iNK tumor
Trafficking
Tumor Antigen expression
Tumor immune microenvironment
Tumor Biology
Tumor burden and
other baseline
biomarkers
PD biomarkers/
B cell aplasia
Cellular
Immunogenicity
Humoral
Immunogenicity
Cytokines:
CRS,
neurotoxicity
ELiPSE-1 translational endpoints |
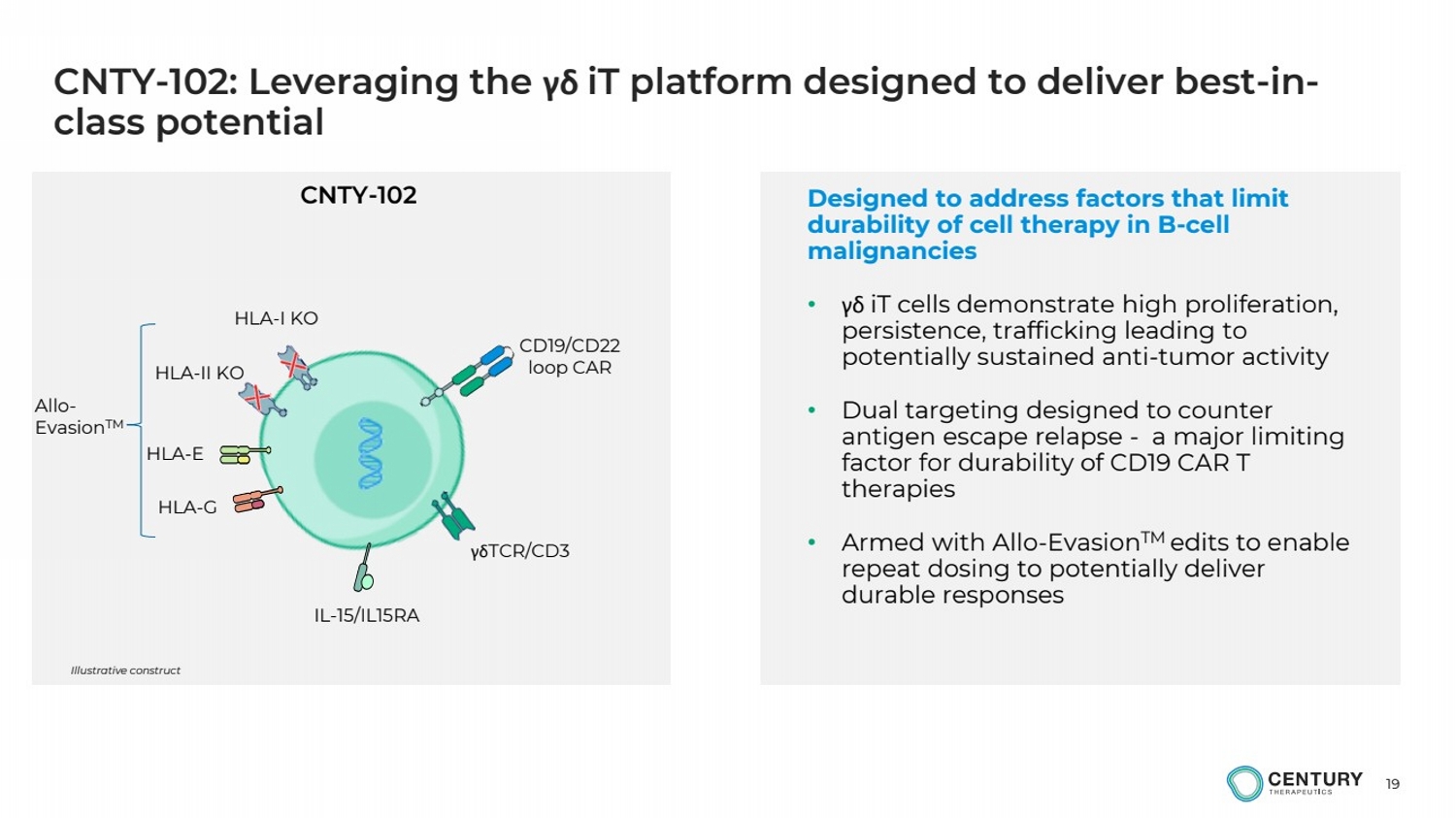
| 19
CNTY-102: Leveraging the γδ iT platform designed to deliver best-in-class potential
HLA-II KO
Illustrative construct
γδTCR/CD3
IL-15/IL15RA
HLA-E
HLA-G
HLA-I KO
CD19/CD22
loop CAR
Designed to address factors that limit
durability of cell therapy in B-cell
malignancies
• γδ iT cells demonstrate high proliferation,
persistence, trafficking leading to
potentially sustained anti-tumor activity
• Dual targeting designed to counter
antigen escape relapse - a major limiting
factor for durability of CD19 CAR T
therapies
• Armed with Allo-EvasionTM edits to enable
repeat dosing to potentially deliver
durable responses
CNTY-102
Allo-EvasionTM |
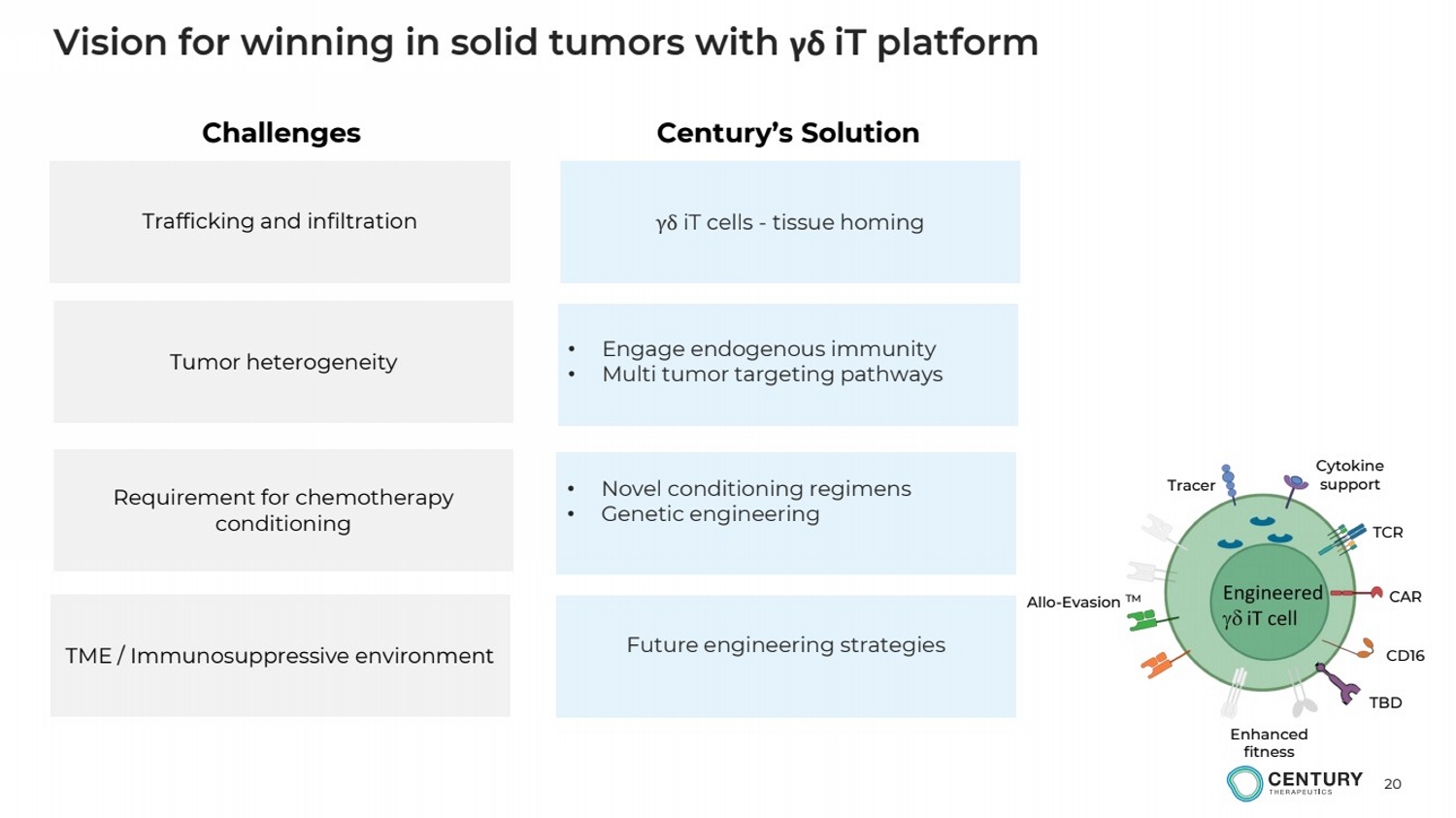
| 20
Vision for winning in solid tumors with γδ iT platform
Trafficking and infiltration γδ iT cells - tissue homing
TME / Immunosuppressive environment
Requirement for chemotherapy
conditioning
Tumor heterogeneity
• Novel conditioning regimens
• Genetic engineering
Challenges Century’s Solution
Future engineering strategies
• Engage endogenous immunity
• Multi tumor targeting pathways
CAR
CD16
TCR
Cytokine
support
Allo-Evasion TM
Enhanced
fitness
Tracer
TBD |
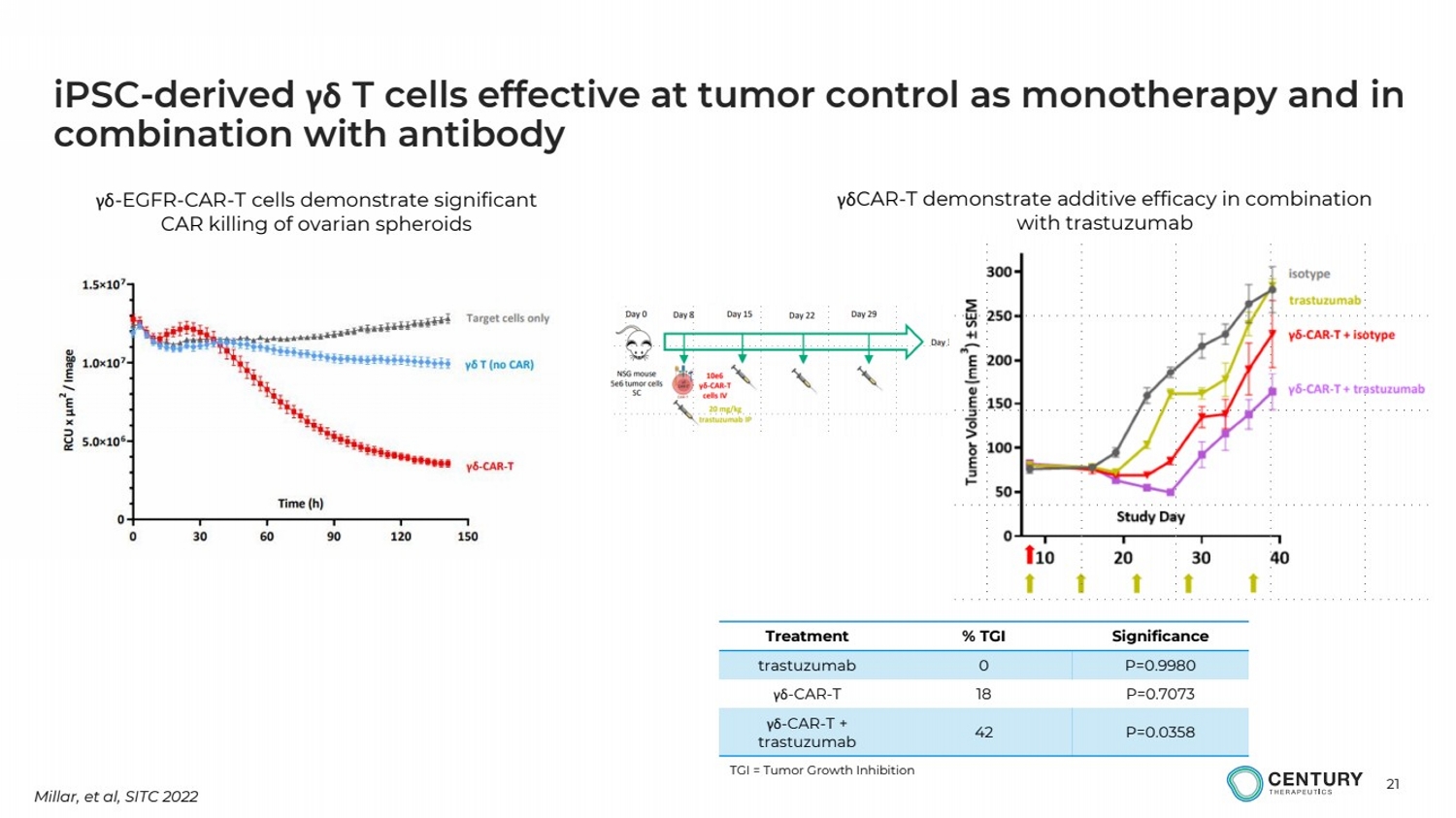
| 21
iPSC-derived γδ T cells effective at tumor control as monotherapy and in
combination with antibody
Millar, et al, SITC 2022
γδ-EGFR-CAR-T cells demonstrate significant
CAR killing of ovarian spheroids
γδCAR-T demonstrate additive efficacy in combination
with trastuzumab
Treatment % TGI Significance
trastuzumab 0 P=0.9980
γδ-CAR-T 18 P=0.7073
γδ-CAR-T +
trastuzumab 42 P=0.0358
TGI = Tumor Growth Inhibition |
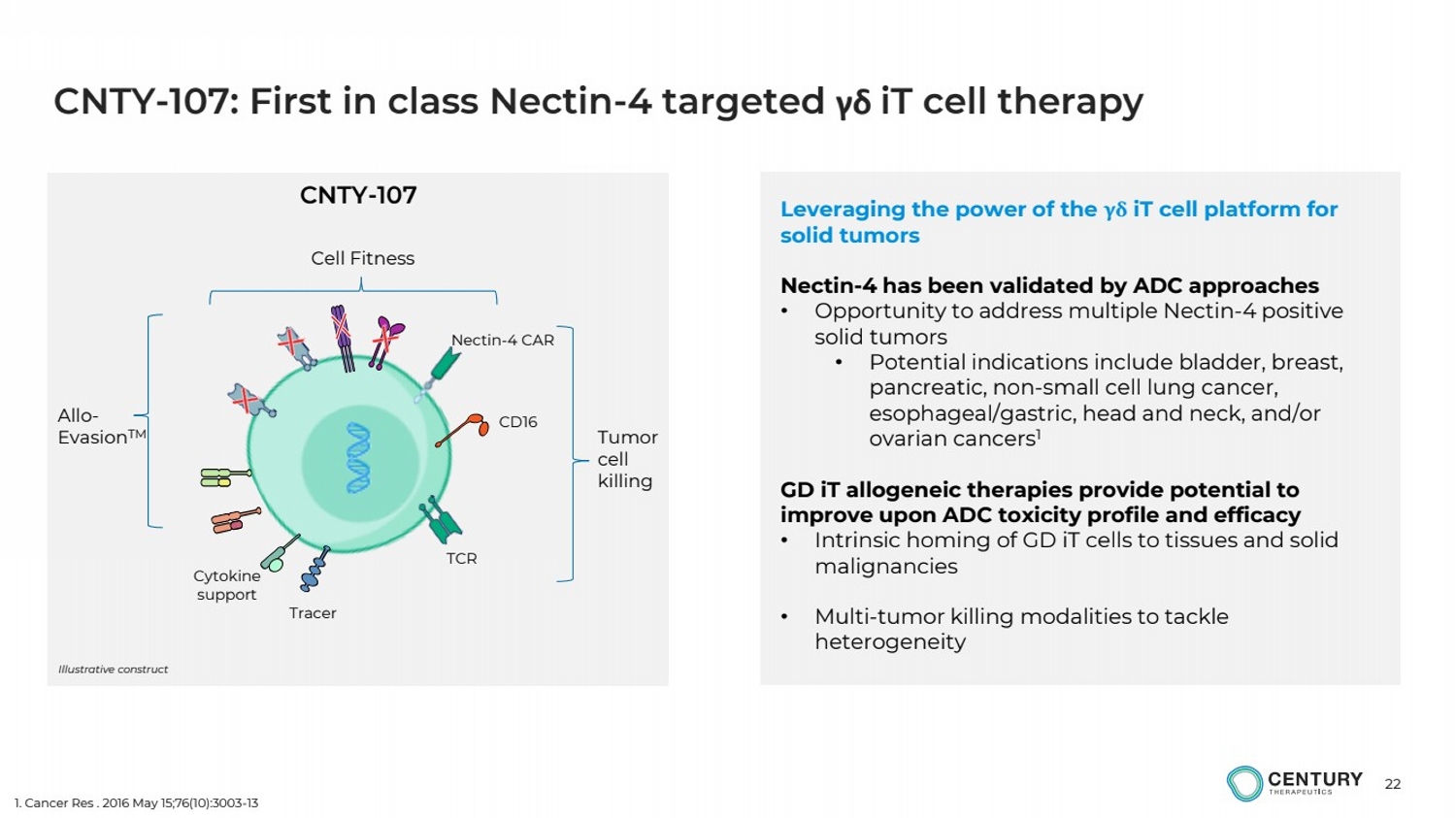
| 22
CNTY-107: First in class Nectin-4 targeted γδ iT cell therapy
Leveraging the power of the γδ iT cell platform for
solid tumors
Nectin-4 has been validated by ADC approaches
• Opportunity to address multiple Nectin-4 positive
solid tumors
• Potential indications include bladder, breast,
pancreatic, non-small cell lung cancer,
esophageal/gastric, head and neck, and/or
ovarian cancers1
GD iT allogeneic therapies provide potential to
improve upon ADC toxicity profile and efficacy
• Intrinsic homing of GD iT cells to tissues and solid
malignancies
• Multi-tumor killing modalities to tackle
heterogeneity
Tumor
cell
killing
Allo-EvasionTM
Cell Fitness
CNTY-107
Illustrative construct
Nectin-4 CAR
CD16
Tracer
TCR
Cytokine
support
1. Cancer Res . 2016 May 15;76(10):3003-13 |

| 23
Investment Thesis
Next generation platforms for iNK and gamma delta iT candidates
Foundational investments in iPSC technology, genetic editing, and manufacturing
Experienced team in R&D, immuno-oncology, manufacturing and
commercialization
Exemplified by FDA clearance of Century’s first IND for CNTY-101 & trial execution
Well capitalized with cash runway into 2026
$301.0M in cash, cash equivalents and investments at the end of 2Q23; operational
efficiencies designed to enable delivery on key milestones, clinical data |

| Emerging leader in cell therapies for cancer
Comprehensive iPSC
cell platform
For immune effector
cells
Technical Expertise
Genetic and protein
engineering, process
development and
immuno-oncology
Foundation in
Science
Continuing investment
in innovation drives R&D
State-of-the-art GMP
manufacturing facility
Fully operational, enabling
improved and faster
product iteration
Financial Strength
Cash runway into 2026,
Ended 2Q23 with cash,
cash equivalents, and
investments of $301M
~165
Employees including
experienced leaders
and entrepreneurs
Emerging pipeline
of candidates
Product engine
anticipated to deliver
additional candidates
and INDs in the coming
years
BMS Discovery
Collaboration
Initial focus on AML
(CNTY-104) and Multiple
Myeloma (CNTY-106) |
| Thank you. |
Cover
|
Aug. 09, 2023 |
| Cover [Abstract] |
|
| Document Type |
8-K
|
| Amendment Flag |
false
|
| Document Period End Date |
Aug. 09, 2023
|
| Entity File Number |
001-40498
|
| Entity Registrant Name |
Century Therapeutics, Inc.
|
| Entity Central Index Key |
0001850119
|
| Entity Tax Identification Number |
84-2040295
|
| Entity Incorporation, State or Country Code |
DE
|
| Entity Address, Address Line One |
25
North 8th Street,
|
| Entity Address, Address Line Two |
11th Floor
|
| Entity Address, City or Town |
Philadelphia
|
| Entity Address, State or Province |
PA
|
| Entity Address, Postal Zip Code |
19104
|
| City Area Code |
267
|
| Local Phone Number |
817-5790
|
| Written Communications |
false
|
| Soliciting Material |
false
|
| Pre-commencement Tender Offer |
false
|
| Pre-commencement Issuer Tender Offer |
false
|
| Title of 12(b) Security |
Common Stock, par value $0.0001 per share
|
| Trading Symbol |
IPSC
|
| Security Exchange Name |
NASDAQ
|
| Entity Emerging Growth Company |
true
|
| Elected Not To Use the Extended Transition Period |
false
|
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 2 such as Street or Suite number
| Name: |
dei_EntityAddressAddressLine2 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the state or province.
| Name: |
dei_EntityAddressStateOrProvince |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:stateOrProvinceItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCommission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.
| Name: |
dei_EntityFileNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fileNumberItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 13e
-Subsection 4c
| Name: |
dei_PreCommencementIssuerTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 14d
-Subsection 2b
| Name: |
dei_PreCommencementTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTitle of a 12(b) registered security. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b
| Name: |
dei_Security12bTitle |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:securityTitleItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the Exchange on which a security is registered. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection d1-1
| Name: |
dei_SecurityExchangeName |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarExchangeCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Section 14a
-Number 240
-Subsection 12
| Name: |
dei_SolicitingMaterial |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:tradingSymbolItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Securities Act
-Number 230
-Section 425
| Name: |
dei_WrittenCommunications |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
Grafico Azioni Century Therapeutics (NASDAQ:IPSC)
Storico
Da Apr 2024 a Mag 2024

Grafico Azioni Century Therapeutics (NASDAQ:IPSC)
Storico
Da Mag 2023 a Mag 2024
