Seqirus Gets FDA Approval for Afluria Quadrivalent in Young Children
23 Ottobre 2018 - 2:58PM
Dow Jones News
By Chris Wack
Seqirus said Tuesday that the U.S. Food and Drug Administration
has approved Afluria Quadrivalent, an influenza vaccine, for use in
people six months of age and older.
According to the Summit-N.J.-based Seqirus, the approval also
applies to the trivalent formulation of Afluria.
Afluria Quadrivalent was first approved in the U.S. in August
2016 for adults aged 18 years and above, and helps protect against
two influenza A strain viruses and two B strain viruses.
Afluria Quadrivalent is available in the U.S. for the 2018-2019
influenza season in prefilled syringes as well as multidose
vials.
Seqirus is a unit of Melbourne, Australia-based CSL Limited.
Write to Chris Wack at chris.wack@wsj.com
(END) Dow Jones Newswires
October 23, 2018 08:43 ET (12:43 GMT)
Copyright (c) 2018 Dow Jones & Company, Inc.
Grafico Azioni CSL (ASX:CSL)
Storico
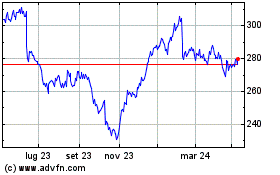
Da Mar 2024 a Apr 2024
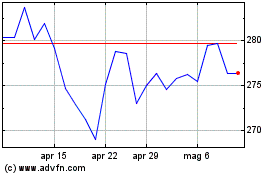
Grafico Azioni CSL (ASX:CSL)
Storico
Da Apr 2023 a Apr 2024