Oxurion NV - Journal of Pharmacokinetics and Pharmacodynamics highlights desirable PK/PD profiles of THR-687 and THR-149
16 Agosto 2021 - 8:00AM

Oxurion NV - Journal of Pharmacokinetics and Pharmacodynamics
highlights desirable PK/PD profiles of THR-687 and THR-149
THR-149 and THR-687 show targeted
activity in the eye following intravitreal
injection with low systemic exposure
Novel pharmacokinetic models developed by
Oxurion may have broad utility to analyze systemic exposure of a
wide variety of drugs delivered in the eye
Leuven, BE, Boston, MA, US – August 16, 2021 – 8.00 AM
CET – Oxurion NV (Euronext Brussels: OXUR), a
biopharmaceutical company developing next generation standard of
care therapies for retinal vascular disorders, announces the
publication of two related papers in the Journal of
Pharmacokinetics and Pharmacodynamics entitled “Systemic
exposure following intravitreal administration of therapeutic
agents: an integrated pharmacokinetic approach.”
These papers describe the pharmacokinetic properties of THR-149
and THR-687 following intravitreal (IVT) injection in animals and
utilize novel pharmacokinetic models developed by Oxurion to
accurately assess the systemic levels of these exciting novel drug
candidates, which are both in clinical development for diabetic
macular edema (DME).THR-149 is a plasma kallikrein inhibitor being
developed as a potential new standard of care for the 40-50% of DME
patients showing suboptimal response to anti-VEGF therapy. With
that, THR-149 holds potential to grow the existing $4.5B DME
market.
THR-687 is a pan-RGD integrin antagonist initially being
developed as a potential first line therapy for treatment of DME
patients. In addition, Oxurion believes that THR-687 could deliver
improved clinical outcomes to the large number of patients with wet
age macular degeneration (AMD) and retinal vein occlusion (RVO)
which together with DME represents a >$12B market
opportunity.
IVT injection remains the preferred route of administration for
pharmacological agents intended for the treatment of back of the
eye diseases such as diabetic macular edema (DME) and neovascular
age-related macular degeneration (wet AMD). The procedure
enables drugs to be delivered locally at high concentrations whilst
limiting systemic exposure and associated risk of systemic adverse
events.
Intravitreally-delivered drugs do, however, enter the general
circulation and achieving an accurate understanding of systemic
exposure as enabled by these new pharmacokinetic models is pivotal
for the evaluation and development of drugs administered in the
eye.
Prof Alan Stitt, Ph.D., Chief Scientific Officer (CSO)
of Oxurion, said, “These publications in the Journal of
Pharmacokinetics and Pharmacodynamics underline the highly
attractive clinical profiles of both THR-149 and THR-687. Both
compounds show a short intravitreal half-life but sustained
biological activity with low systemic exposure due to their rapid
clearance. This enables targeted activity in the eye combined with
minimal risk of systemic side effects. The animal models and
analytical techniques that we have developed have proven accurate
in predicting circulating plasma levels of these novel drug
candidates following intravitreal injection, and potentially have
broad predictive value for a variety of therapeutics administered
by IVT. These publications reflect the calibre and thought
leadership of the high-quality science that we are generating at
Oxurion.”
Articles can be accessed at:
https://link.springer.com/article/10.1007/s10928-021-09773-w
https://link.springer.com/article/10.1007/s10928-021-09774-9
END
For further information please contact:
|
Oxurion NVWouter Piepers,Global Head of Investor Relations&
Corporate CommunicationsTel: +32 478 33 56
32wouter.piepers@oxurion.com - |
EUMEDiSTRAVA ConsultingDavid Dible/ Sylvie Berrebi/Frazer HallTel:
+44 20 7638 9571oxurion@medistrava.com -USWestwicke, an ICR
Company Christopher BrinzeyTel: +1 617 835
9304chris.brinzey@westwicke.com |
About Oxurion
Oxurion (Euronext Brussels: OXUR) is a biopharmaceutical company
developing next generation standard of care ophthalmic therapies,
which are designed to better preserve vision in patients with
retinal vascular disorders including diabetic macular edema (DME),
the leading cause of vision loss in diabetic patients worldwide as
well as other conditions, including wet age-related macular
degeneration (AMD) and retinal vein occlusion (RVO).
Oxurion is aiming to build a leading global franchise in the
treatment of retinal vascular disorders based on the successful
development of its two novel therapeutics:
- THR-687 is a pan-RGD
integrin antagonist that is initially being developed as a
potential first line therapy for DME patients. Positive topline
results in a Phase 1 clinical study assessing THR-687 as a
treatment for DME were announced in 2020. Oxurion is currently
conducting a Phase 2 clinical trial evaluating THR-687 in patients
with DME. THR-687 also has the potential to deliver improved
treatment outcomes for patients with wet AMD and RVO.
- THR-149 is a plasma
kallikrein inhibitor being developed as a potential new standard of
care for the 40-50% of DME patients showing suboptimal response to
anti-VEGF therapy. THR-149 has shown positive topline Phase 1
results for the treatment of DME. The company is currently
conducting a Phase 2 clinical trial evaluating multiple injections
of THR-149 in DME patients previously showing suboptimal response
to anti-VEGF therapy. Dose selection data from Part A of the study,
which is fully enrolled, is expected in the second half of
2021.
Oxurion is headquartered in Leuven, Belgium, and is listed on
the Euronext Brussels exchange under the symbol OXUR. More
information is available at www.oxurion.com.
Important information about forward-looking
statements
Certain statements in this press release may be considered
“forward-looking”. Such forward-looking statements are based on
current expectations, and, accordingly, entail and are influenced
by various risks and uncertainties. The Company therefore cannot
provide any assurance that such forward-looking statements will
materialize and does not assume an obligation to update or revise
any forward-looking statement, whether as a result of new
information, future events, or any other reason. Additional
information concerning risks and uncertainties affecting the
business and other factors that could cause actual results to
differ materially from any forward-looking statement is contained
in the Company’s Annual Report. This press release does not
constitute an offer or invitation for the sale or purchase of
securities or assets of Oxurion in any jurisdiction. No
securities of Oxurion may be offered or sold within the United
States without registration under the U.S. Securities Act of 1933,
as amended, or in compliance with an exemption therefrom, and in
accordance with any applicable U.S. state securities laws.
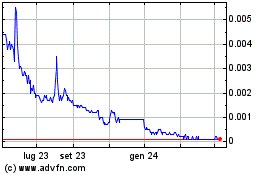
Grafico Azioni Oxurion NV (EU:OXUR)
Storico
Da Mar 2024 a Apr 2024
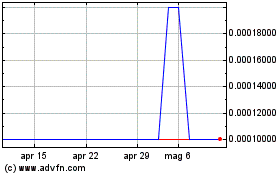
Grafico Azioni Oxurion NV (EU:OXUR)
Storico
Da Apr 2023 a Apr 2024