Valneva Switches Focus to Bilateral Discussions to Supply its Inactivated, Adjuvanted COVID-19 Vaccine Candidate VLA2001
20 Aprile 2021 - 11:59PM

Valneva Switches Focus to Bilateral Discussions to Supply its
Inactivated, Adjuvanted COVID-19 Vaccine Candidate VLA2001
Saint-Herblain
(France), April
20, 2021 – Valneva SE, a
specialty vaccine company focused on the development and
commercialization of prophylactic vaccines for infectious diseases
with significant unmet medical need, today announced it is now
focusing on bilateral discussions, on a country by country basis,
to supply its inactivated, adjuvanted COVID-19 vaccine candidate,
VLA2001, and is consequently deprioritizing the ongoing centralized
discussions with the European Commission (EC).
This follows the recent announcement on April 6,
2021 of positive initial results from the Phase 1/2 clinical trial
of VLA20011.
Valneva previously announced the advanced stage
of its discussions, which started in the third quarter of 2020,
with the EC on January 12, 20212. In September 2020, Valneva
announced a collaboration with the UK government to provide up to
190 million doses of VLA2001 through 20253, of which 100 million
doses have already been ordered.
Thomas
Lingelbach, Chief Executive Officer of
Valneva, said, “We’ve committed significant time and
effort to try to meet the needs of the central EC procurement
process. Despite our recent clinical data, we have not made
meaningful progress and have not yet secured a supply agreement. We
are therefore now focusing our efforts on those European Union
member states, and interested parties outside the EU, who would
like to include our inactivated approach within their vaccination
strategy. We are convinced that VLA2001 has an important role to
play in the future, including boosters or potential modifications
to the vaccine to address variants”.
Valneva has the only inactivated vaccine
candidate in clinical trials against COVID-19 in Europe and is
preparing a pivotal, comparative immunogenicity Phase 3 clinical
trial expected to commence by the end of April 2021 with the aim of
making regulatory licensure submissions in the autumn of 2021.
About the Novel Coronavirus SARS-CoV-2
and COVID-19 DiseaseSARS-CoV-2 is a new coronavirus
identified in late 2019 and belongs to a family of enveloped RNA
viruses that include MERS and SARS, both of which caused serious
human infections of the respiratory system. The virus, which causes
a disease named COVID-19, has never before been found in humans.
Since this outbreak was first reported, the virus has caused
millions of deaths globally4. It has been declared a pandemic by
the World Health Organization (WHO).
About VLA2001VLA2001 is
currently the only whole virus, inactivated, adjuvanted vaccine
candidate in clinical trials against COVID-19 in Europe. It is
intended for active immunization of at-risk populations to prevent
carriage and symptomatic infection with COVID-19 during the ongoing
pandemic and potentially later for routine vaccination including
addressing new variants. VLA2001 may also be suited for boosting,
as repeat booster vaccinations have been shown to work well with
whole virus inactivated vaccines. VLA2001 is produced on Valneva’s
established Vero-cell platform, leveraging the manufacturing
technology for Valneva’s licensed Japanese encephalitis vaccine,
IXIARO®. VLA2001 consists of inactivated whole virus particles of
SARS-CoV-2 with high S-protein density, in combination with two
adjuvants, alum and CpG 1018. This adjuvant combination has
consistently induced higher antibody levels in preclinical
experiments than alum-only formulations and shown a shift of the
immune response towards Th1. CpG 1018 adjuvant, supplied by Dynavax
Technologies Corporation (Nasdaq: DVAX), is a component of the US
FDA- and EMA-approved HEPLISAV-B® vaccine. The manufacturing
process for VLA2001, which has already been upscaled to final
industrial scale, includes inactivation with BPL to preserve the
native structure of the S-protein. VLA2001 is expected to conform
with standard cold chain requirements (2 degrees to 8 degrees
Celsius).
About Valneva SEValneva is a
specialty vaccine company focused on the development and
commercialization of prophylactic vaccines for infectious diseases
with significant unmet medical need. We take a highly specialized
and targeted approach to vaccine development, beginning with the
identification of deadly and debilitating infectious diseases that
lack a prophylactic vaccine solution and for which there are
limited therapeutic treatment options. We then apply our deep
understanding of vaccine science, including our expertise across
multiple vaccine modalities, as well as our established vaccine
development capabilities, to develop prophylactic vaccines to
address these diseases. We have leveraged our expertise and
capabilities both to successfully commercialize two vaccines and to
rapidly advance a broad range of vaccine candidates into and
through the clinic, including candidates against Lyme disease, the
chikungunya virus and COVID-19.
|
Valneva Investor and Media ContactsLaetitia
Bachelot-FontaineDirector Investor Relations & Corporate
CommunicationsM +33 (0)6 4516
7099investors@valneva.com |
Dan
SharpGovernment & Public Affairs Manager T
+44-(0)7436-244309communications@valneva.com |
|
|
Valneva Forward-Looking
StatementsThis press release contains certain
forward-looking statements relating to the business of Valneva,
including with respect to the progress, timing, results and
completion of research, development and clinical trials for product
candidates, timing and volume expectations with respect to the
manufacture of our product candidates; the ability to market,
commercialize and achieve market acceptance for product candidates,
the ability to protect intellectual property and operate the
business without infringing on the intellectual property rights of
others, estimates for future performance and estimates regarding
anticipated operating losses, future revenues, capital requirements
and needs for additional financing. In addition, even if the actual
results or development of Valneva are consistent with the
forward-looking statements contained in this press release, those
results or developments of Valneva may not be sustained in the
future. In some cases, you can identify forward-looking statements
by words such as "could," "should," "may," "expects,"
"anticipates," "believes," "intends," "estimates," "aims,"
"targets," or similar words. These forward-looking statements are
based largely on the current expectations of Valneva as of the date
of this press release and are subject to a number of known and
unknown risks and uncertainties and other factors that may cause
actual results, performance or achievements to be materially
different from any future results, performance or achievement
expressed or implied by these forward-looking statements. In
particular, the expectations of Valneva could be affected by, among
other things, uncertainties involved in the development and
manufacture of vaccines, unexpected clinical trial results,
unexpected regulatory actions or delays, competition in general,
currency fluctuations, the impact of the global and European credit
crisis, and the ability to obtain or maintain patent or other
proprietary intellectual property protection. Success in
preclinical studies or earlier clinical trials may not be
indicative of results in future clinical trials. In light of these
risks and uncertainties, there can be no assurance that the
forward-looking statements made during this presentation will in
fact be realized. Valneva is providing the information in these
materials as of this press release, and disclaim any intention or
obligation to publicly update or revise any forward-looking
statements, whether as a result of new information, future events,
or otherwise.
1 Valneva Reports Positive Phase 1/2 Data for Its Inactivated,
Adjuvanted COVID-19 Vaccine Candidate, VLA20012 Valneva in Advanced
Discussions with European Commission to Supply up to 60 Million
Doses of Inactivated, Adjuvanted COVID-19 Vaccine Candidate3
Valneva Announces Major COVID-19 Vaccine Partnership with U.K.
Government4 https://www.worldometers.info/coronavirus/
- 2021_04_20_VLA2001_EC_PR_EN_Final
Grafico Azioni Valneva (EU:VLA)
Storico
Da Mar 2024 a Apr 2024
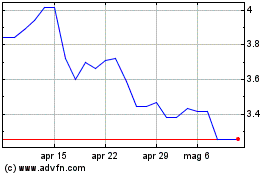
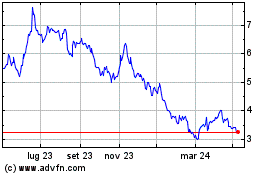
Grafico Azioni Valneva (EU:VLA)
Storico
Da Apr 2023 a Apr 2024