MedinCell’s Long-Acting Injectable to Fight Malaria Ready to Enter Regulatory Development
14 Giugno 2021 - 7:14PM
Business Wire
MedinCell and the project consortium members, IRD, IRSS and
CIRDES, have successfully designed, tested, and confirmed with
Unitaid the selection of the lead formulation. Regulatory
preclinical activities are starting with the objective of a first
clinical trial in 2023.
MedinCell and the consortium members have conducted an in
vivo proof of concept on cattle that showed a 3-month mosquitocidal
effectiveness of an injectable long-acting formulation of
Ivermectin based on the proprietary BEPO® technology.
The investigational product mdc-STM aims at reducing the
transmission of the parasite responsible for Malaria, thanks to a
killing effect on the vector mosquitoes when they bite treated
people.
Malaria remains one of the main health threats worldwide with
more than 200 million people infected yearly.
In March 2020, global health agency Unitaid granted MedinCell
with a $6.4 million subvention over three years to conduct the
formulation and preclinical activities of the program.
mdc-STM benefits from synergies with other MedinCell’s
programs based on Ivermectin all of them using different
formulation and doses fitted to their specific indications.
Access the complete press release.
About MedinCell
MedinCell is a clinical stage pharmaceutical company that
develops a portfolio of long-acting injectable products in various
therapeutic areas by combining its proprietary BEPO® technology
with active ingredients already known and marketed. Through the
controlled and extended release of the active pharmaceutical
ingredient, MedinCell makes medical treatments more efficient,
particularly thanks to improved compliance, i.e. compliance with
medical prescriptions, and to a significant reduction in the
quantity of medication required as part of a one-off or chronic
treatment. The BEPO® technology makes it possible to control and
guarantee the regular delivery of a drug at the optimal therapeutic
dose for several days, weeks or months starting from the
subcutaneous or local injection of a simple deposit of a few
millimeters, fully bioresorbable. Based in Montpellier, MedinCell
currently employs more than 140 people representing over 25
different nationalities.
www.medincell.com
View source
version on businesswire.com: https://www.businesswire.com/news/home/20210614005689/en/
MedinCell David Heuzé Head of communication
david.heuze@medincell.com +33 (0)6 83 25 21 86
NewCap Louis-Victor Delouvrier Relations investisseurs
medincell@newcap.eu +33 (0)1 44 71 94 94
NewCap Nicolas Merigeau Media Relations medincell@newcap.eu +33
(0)1 44 71 94 94
Grafico Azioni Medincell (EU:MEDCL)
Storico
Da Mar 2024 a Apr 2024
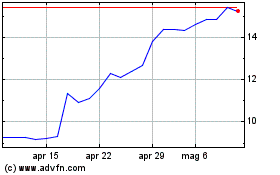
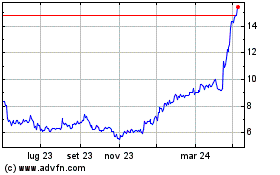
Grafico Azioni Medincell (EU:MEDCL)
Storico
Da Apr 2023 a Apr 2024